Advertisements
Advertisements
Question
Mention the number of unpaired electrons and geometry of the following complex:
\[\ce{[Ni(CN)4]^2-}\]
Advertisements
Solution
The oxidation state of Nickel is +2.
Valence shell electronic configuration of \[\ce{Ni^{2+}}\]
![]()
The complex is square planar geometry, so \[\ce{Ni^{2+}}\] ion uses dsp2 hybrid orbitals.
3d electrons are paired prior to hybridization, and the electronic configuration of \[\ce{Ni^{2+}}\] becomes: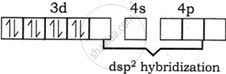
The complex has no unpaired electrons, and hence it is diamagnetic.
APPEARS IN
RELATED QUESTIONS
Answer in brief.
[CoCl4]2− is a tetrahedral complex. Draw its box orbital diagram. State which orbitals participate in hybridization.
Answer in brief.
[CoCl4]2- is a tetrahedral complex. Draw its box orbital diagram. State which orbitals participate in hybridization.
Answer in brief.
What are strong field and weak field ligands ? Give one example of each.
Answer the following question.
Give valence bond description for the bonding in the complex [VCl4]-. Draw box diagrams for the free metal ion. Which hybrid orbitals are used by the metal? State the number of unpaired electrons.
Answer the following with respect to [CoF6]3– ion
- Type of hybridization
- Number of unpaired electrons
- Geometry of complex ion
- Magnetic property
Identify the number of donor groups present in EDTA.
Chlorophyll and haemoglobin are complexes of ____________ respectively.
The INCORRECT match for complex with its geometry is:
What is the value of primary valence of Co in CoCl3?
What is the type of magnetic behavior and geometry respectively in Cuproammonium sulphate (Atomic number of Cu = 29)?
Identify the increasing order of effective magnetic moment of the following elements in their +2 oxidation state.
[Fe (Z = 26), Co (Z = 27), Ni (Z = 28), Cu (Z = 29)]
Which of the following types of square planar complexes can show geometrical isomerism [M = metal, a, b, = monodentate ligand]?
Explain the formation of [CoF6]3Θ complex with respect to
- Hybridisation
- Magnetic properties
- Inner/outer complex
- Geometry
Describe the bonding in the tetrahedral complex Ni(CO)4 on the basis of valence bond theory. Give the orbital diagrams of metal atoms in free state and in the complex. Mention the number of unpaired electrons in the complex.
A compound forms hcp structure. What is the number of (a) octahedral voids, (b) tetrahedral voids, (c) total voids formed in 0.2 mol of the compound?
Give VBT description of the bonding in a square planar complex [Cu(NH3) 4]2+. Show orbital diagrams for free metal ion and metal ion in the complex. Which hybrid orbitals are used by metal for bonding? State magnetic nature of the complex.
A compound forms a hep structure. Calculate the number of octahedral voids in 0.4 mol. (NA = 6.022 × 1023).
A compound forms a hep structure. Calculate the number of octahedral voids in 0.4 mol. (NA = 6.022 × 1023 )
Octahedral complex have ______ hybridisation.
Give the limitations of VBT.
A compound forms hexagonal close packed (hcp) structure. What is the number of (i) Octahedral voids, (ii) Tetrahedral voids, (iii) Total voids formed in 0.7 mol of it.
Mention the type of hybridization in [Co(NH3)6]3+ complex.
