Advertisements
Advertisements
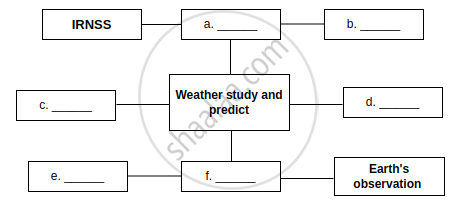
Complete the Following table.
| Sr. no. | Type of Satellite | The names of Indian Satellite and launcher |
| (1) | Navigational Satellite | Satellite: ______ |
| Launcher: ______ | ||
| (2) | Earth Observation Satellite | Satellite: ______ |
| Launcher: ______ |
Concept: Orbits of Artificial Satellites
Complete the following equations:
Concept: Orbits of Artificial Satellites
What is meant by a satellite launch vehicle?
Concept: Satellite Launch Vehicles
Name any one Indian satellite launch vehicle.
Concept: Satellite Launch Vehicles
The orbit of a satellite is exactly 35780 km above the earth's surface and its tangential velocity is 3.08 km/s.
How much time the satellite will take to complete one revolution around the earth?
(Radius of earth = 6400 km.)
Concept: Orbits of Artificial Satellites
Fill in the blank:
The modern periodic table consists of ……………. periods.
Concept: The Modern Periodic Table
Fill in the blank:
The formulae of chloride of metal M is MCl2. The metal M belongs to ……………. group.
Concept: Periods and Electronic Configuration
The element eka-silicon in Mendeleev’s periodic table is known as ………… in the modern periodic table.
Concept: Mendeleev’s Periodic Table >> Insights into Mendeleev’s Periodic Table
The element eka-silicon in Mendeleev’s periodic table is known as ………… in the modern periodic table.
Concept: Mendeleev’s Periodic Table >> Insights into Mendeleev’s Periodic Table
Elements in the same group show the same valency. Give scientific reason.
Concept: Periods and Electronic Configuration
Write the electronic configuration of K and Ne.
Concept: Periods and Electronic Configuration
The atomic masses of three elements A, B and C having similar chemical properties are 7, 23 and 39, respectively.
- Calculate the average atomic mass of elements A and C.
- Compare the average atomic mass with atomic mass of B.
- What could the elements A, B and C be?
Concept: Periods and Electronic Configuration
Classify the following elements into metals, non-metals and metalliods
As, C, Hg, Mg, S, Si
Concept: Periods and Electronic Configuration
Write the merits of the modern periodic table over Mendeleev’s Periodic table
Concept: The Modern Periodic Table
Differentiate between Normal elements and Transition elements.
Concept: Periods and Electronic Configuration
What would be the correct order if Zn, Fe, Al and Cu are arranged in increasing order of reactivity?
Concept: The Modern Periodic Table
Atomic number of metal ‚A‛ is 11, while atomic number of metal ‚B‛ is 20. Which of them will be more reactive? Write the chemical reaction of dilute HCl with metal ‚A‛.
Concept: Periods and Electronic Configuration
Taking into consideration the period of the elements given below, answer the following questions:
| Elements | Atomic Radius (pm) |
| O | 66 |
| B | 88 |
| C | 77 |
| N | 74 |
| Be | 111 |
| Li | 152 |
- Arrange the above elements in decreasing order of their atomic radii.
- State the period to which the above elements belong.
- Why this arrangement of elements is similar to the above period of modern periodic table?
- Which of the above elements have the biggest and the smallest atom?
- What is the periodic trend observed in the variation of atomic radius while going from left to right within a period?
Concept: Periods and Electronic Configuration
Fill in the blank and rewrite the completed statement:
________ group in the periodic table contains elements that are all gases at room temperature.
Concept: Periods and Electronic Configuration
Concept: Periods and Electronic Configuration
