Advertisements
Advertisements
प्रश्न
Write any ‘two’ uses of terylene.
Mention some of the important uses of the terylene polymer.
Advertisements
उत्तर
- It is widely used to make textile fibres for clothing, sportswear, and upholstery.
- It is used to manufacture plastic bottles, films, and packaging containers.
- It serves in industrial products such as ropes, tyre‑cords, and conveyor belts.
- It is common in home furnishings like curtains, beddings, and upholstery.
- It is used in automotive and composite parts for interiors and panels.
- Recycled Terylene is used for medical textiles and geotextiles.
Notes
Students should refer to the answer according to their questions.
APPEARS IN
संबंधित प्रश्न
Bakelite is the polymer of:
(a) Benzaldchyde and phenol
(b) Acetaldehyde and phenol
(c) Formaldehyde and phenol
(d) Formaldehyde and benzyl alcohol
Answer the following in one sentence.
What type of intermolecular force leads to high-density polymer?
Answer the following.
Name the polymer type in which following linkage is present.
\[\begin{array}{cc}\ce{- C - O -}\\||\phantom{.....}\\
\ce{O\phantom{.....}}\end{array}\]
Answer the following.
Write name and formula of raw material from which bakelite is made.
Identify condensation polymers and addition polymers from the following.
-(CO(CH2)4 - CONH(CH2)6NH-)n
Identify condensation polymers and addition polymers from the following.
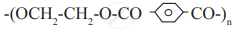
Attempt the following:
Write the chemical reactions involved in the manufacture of Nylon 6,6.
Attempt the following:
Explain the vulcanisation of rubber. Which vulcanizing agents are used for the following synthetic rubber?
a. Neoprene
b. Buna-N
Attempt the following:
What is meant by LDP and HDP? Mention the basic difference between the same with suitable examples.
Answer the following.
Write main specialities of Buna-S, Neoprene rubber?
Write the structure of isoprene and the polymer obtained from it.
Monomer used for preparation of polyacrylonitrile is _____________
Write chemical reaction for preparation of the following.
Neoprene
Write the name of one example of each polymer in which following repeating units.
\[\begin{array}{cc}
\ce{(-CF2-CF2-), -[NH-(CH2)5-CO] -, -(CH2-CH-), (-CH2-CH2-)}\\
\phantom{............................}|\\
\phantom{..............................}\ce{CN}
\end{array}\]
Write the name and formulae of the monomers used for the preparation of dacron.
Write two uses and two properties of polythene.
Which among the following polymers is used for making handles of cooker?
Which of the following is the monomer of neoprene?
Identify the polymer obtained by polymerization of n moles of acrylonitrile.
Which of the following catalysts is used in preparation of terylene?
Which of the following compounds is used to prepare orlon?
Which among the following monomers is used to prepare Teflon?
Which of the following pair of compounds is used as monomers for bakelite?
Which of the following polymers is used as insulation for cables?
Which of the following is not a semisynthetic polymer?
Which of the following polymers, need atleast one diene monomer for their preparation?
(i) Dacron
(ii) Buna-S
(iii) Neoprene
(iv) Novolac
Name the polymers used in laminated sheets and give the name of monomeric units involved in its formation.
Match the polymers given in Column I with the type of linkage present in them given in Column II.
| Column I | Column II |
| (i) Terylene | (a) Glycosidic linkage |
| (ii) Nylon | (b) Ester linkage |
| (iii) Cellulose | (c) Phosphodiester linkage |
| (iv) Protein | (d) Amide linkage |
| v) RNA |
Which of the following products is formed when benzaldehyde is treated with CH3MgBr and the addition product so obtained is subjected to acid hydrolysis?
The monomer of Teflon is ______.
F2C = CF2 is monomer of the polymer -
Which of the following polymer has ester linkage?
Which of the following is an example of polyester?
Nylon threads are made of ______.
Which of the following polymers do not involve cross linkages?
Which of the following is a polymer of enzyme?
Which of the foolowing polymer is used in the manufacture of insulators.
Answer the following.
Write the structure of isoprene and the polymer obtained from it.
Answer the following.
Write the structure of isoprene and the polymer obtained from it.
Write the structure of isoprene and the polymer obtained from it.
Write the structure of isoprene and the polymer obtained from it.
Name and draw structure of the repeating unit in natural rubber.
Write the structure and name of monomer of Natural rubber.
Write the structure of isoprene and the polymer obtained from it.
Name and draw structure of the repeating unit in natural rubber.
Write the structure of isoprene and the polymer obtained from it.
Name and draw structure of the repeating unit in natural rubber.
Write the structure of isoprene and the polymer obtained from it.
Name and draw structure of the repeating unit in natural rubber.
Write the structure of isoprene and the polymer obtained from it.
Name and draw the structure of the repeating unit in natural rubber.
Which from following statements is NOT true regarding neoprene?
