Advertisements
Advertisements
प्रश्न
Attempt the following:
Write the chemical reactions involved in the manufacture of Nylon 6,6.
Advertisements
उत्तर

APPEARS IN
संबंधित प्रश्न
Write the reactions involved in the preparation of PVC
Write the chemical reaction to prepare novolac polymer.
Explain the following term: Homopolymers
Bakelite is the polymer of:
(a) Benzaldchyde and phenol
(b) Acetaldehyde and phenol
(c) Formaldehyde and phenol
(d) Formaldehyde and benzyl alcohol
The Zieglar-Natta catalyst is used in the preparation of _______.
(A) LDPE
(B) PHBV
(C) PAN
(D) HDPE
Write the monomers of the following polymer :

Answer the following in one sentence.
Define the term: Vulcanization
Answer the following in one sentence.
What type of intermolecular force leads to high-density polymer?
Answer the following.
Write the reaction of the formation of Terylene.
Answer the following.
Draw the structures of polymers formed from the following monomers
H2N–(CH2)5 – COOH
Name and draw structure of the repeating unit in natural rubber.
Identify condensation polymers and addition polymers from the following.
-(CH2 - CH = CH - CH2 -)n
Identify condensation polymers and addition polymers from the following.
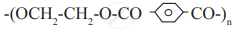
Write the reaction involved in the formation of bakelite.
Nylon 6, 6 is a condensation polymer of hexamethylenediamine and _____________
Monomers ethylene glycol and terephthalic acid undergo condensation polymerization to give polymer called ___________
Monomer of natural rubber is __________
Mention two uses of LDP.
Write chemical reactions for the preparation of high-density polythene.
Define rubber.
Explain the reactions involved in the preparation of viscose rayon.
Which of the following is used as a substitute for wool?
Which among the following polymers is used for making handles of cooker?
Which of the following polymer is used in paints?
Identify the CORRECT statement regarding the following polymer.
\[\begin{array}{cc}
\phantom{....}\ce{O}\phantom{............}\ce{O}\phantom{...................}\ce{H}\phantom{.....}\\
\phantom{....}||\phantom{.............}||\phantom{...................}|\phantom{......}\\
\ce{-[C - (CH2)4 - C - NH - (CH2)6 - N -]_{{n}}}
\end{array}\]
Identify the INCORRECT match.
Which of the following polymers is obtained from chloroprene?
Novolac is obtained from ____________.
Which among the following catalysts is used in the preparation of dacron?
Identify the catalyst used in the manufacture of high density polythene.
Which among the following monomers is used to prepare Teflon?
Which of the following polymers is prepared by using phenol?
Which of the following monomers is used in the manufacture of Neoprene rubber?
Which among the following polymers is used to manufacture chemical containers?
Name the polymers used in laminated sheets and give the name of monomeric units involved in its formation.
Trans - form of poly isoprene is:-
Which one of the following polymers are prepared by addition polymerization?
Which of the following is a polymer of enzyme?
Which of the foolowing polymer is used in the manufacture of insulators.
Name and draw the structure of the repeating unit in natural rubber.
Write the structure of isoprene and the polymer obtained from it.
Write the preparation of viscose rayon.
Write the structure of isoprene and the polymer obtained from it.
Write the name of the monomer of natural rubber.
Another name of terylene is ______.
Name and draw structure of the repeating unit in natural rubber.
Match the following pairs:
| Polymer | Monomer | ||
| (i) | Teflon | (a) | CH2 = CH2 |
| (ii) | PVC | (b) | CF2 = CF2 |
| (iii) | Natural rubber | (c) | Isoprene |
| (iv) | Polythene | (d) | H2C=CHCl |
Write the structure of isoprene and the polymer obtained from it.
Name and draw structure of the repeating unit in natural rubber.
Write the structure of isoprene and the polymer obtained from it.
Write the structure of isoprene and the polymer obtained from it.
Write the structure of isoprene and the polymer obtained from it.
