Advertisements
Advertisements
प्रश्न
Which of the following compounds is NOT prepared by the action of alcoholic NI3 on alkyl halide?
(a) CH3NH2
(b) CH3- CH2- NH2
(c) CH3 - CH2 - CH2 - NH2
(d) (CH3)3 C- NH2
Advertisements
उत्तर
(CH3)3C – NH2
APPEARS IN
संबंधित प्रश्न
What is metamerism?
Write the IUPAC name of the given compound:

Name the following compound according to the IUPAC system.
\[\begin{array}{cc}
\phantom{..................}\ce{CH2OH}\\
\phantom{.........}|\\
\ce{CH3 - CH2 - CH - CH - CH - CH3}\\
\phantom{.........}|\phantom{...................}|\\
\phantom{.............}\ce{CH2Cl}\phantom{..........}\ce{CH3}\phantom{}
\end{array}\]
Name the following compound according to the IUPAC system.
\[\begin{array}{cc}
\ce{CH3 - C = C - CH2OH}\\
|\phantom{......}|\phantom{.....}\\
\ce{CH3}\phantom{.}\ce{Br}\phantom{....}\\
\end{array}\]
Write the IUPAC name of the following compound:
\[\begin{array}{cc}
\phantom{........................}\ce{CH3}\\
\phantom{....................}|\\
\ce{CH3 - CH - CH - C - CH3}\\
|\phantom{.........}|\phantom{.........}|\\
\phantom{...}\ce{CH3}\phantom{....}\ce{OH}\phantom{....}\ce{CH3}\\
\end{array}\]
Write the IUPAC name of the following compound:
\[\begin{array}{cc}
\ce{HO - CH2 - CH - CH2 - OH}\\
|\phantom{...}\\
\ce{OH}\phantom{.}\\
\end{array}\]
Write the IUPAC name of the following compound:

Write the IUPAC name of the following compound:
C6H5 – O – C2H5
Give the IUPAC name of the following ether:
How is phenol converted into the following?
benzoquinone
What.will be the product fonned when chlorobenzene is heated with sodium metal in the presence of dry ether?
Write structural formulae for 1-Ethylcyclohexanol.
Write IUPAC name of the following

In a carbinol system of nomenclature tert.butyl alcohol is named as _______________
Give IUPAC names of the following compound:

When ethyl alcohol reacts with acetic acid, the products formed are:
The IUPAC name of the ether CH2 = CH–CH2OCH3 is:
The heating of phenyl methyl ether with HI produces:
\[\ce{Phenol ->[Zn, dust] 'X' ->[CH3Cl][Anhy. AlCl3] 'Y' ->[Alkaline][KMnO4] 'Z'}\]
The product ‘Z’ is:
Match the structures of the compounds given in Column I with the name of the compounds given in Column II.
| Column I | Column II | |
| (i) |  |
(a) Hydroquinone |
| (ii) |  |
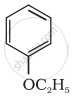
(b) Phenetole |
| (iii) | 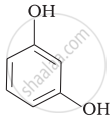 |
(c) Catechol |
| (iv) |  |
(d) o-Cresol |
| (v) |  |
(e) guinone |
| (vi) | 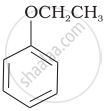 |
(f) Resorcinol |
| (g) Anisole |
Assertion: Addition reaction of water to but-1-ene in acidic medium yields butan-1-ol.
Reason: Addition of water in acidic medium proceeds through the formation of primary carbocation.
Assertion: IUPAC name of the compound
\[\begin{array}{cc}
\ce{CH3 - CH - O - CH2 - CH2 - CH3}\\
|\phantom{....................}\\
\ce{CH3}\phantom{.................}
\end{array}\] is 2-Ethoxy-2-methylethane.
Reason: In IUPAC nomenclature, ether is regarded as hydrocarbon derivative in which a hydrogen atom is replaced by —OR or —OAr group [where R = alkyl group and Ar = aryl group]
Write complete reaction for the bromination of phenol in aqueous and non-aqueous medium.
Write the IUPAC name of the following compound.

Draw structure of the following compound.
2-Methoxypropane
Draw structure of the following compound.
Prop-2-en-1-ol
