Advertisements
Advertisements
प्रश्न
When a metal plate is exposed to a monochromatic beam of light of wavelength 400 nm, a negative potential of 1.1 V is needed to stop the photo current. Find the threshold wavelength for the metal.
(Use h = 6.63 × 10-34J-s = 4.14 × 10-15 eV-s, c = 3 × 108 m/s and me = 9.1 × 10-31kg)
Advertisements
उत्तर
Given :-
Wavelength of light, `λ = 400 "nm" = 400 xx 10^-9 "m"`
Stopping potential, `V_0 = 1.1 V`
From Einstein's photoelectric equation,
`(hc)/λ = (hc)/(λ_0) + eV_0`,
where h = Planck's constant
c = speed of light
λ = wavelength of light
`λ_0` = threshold wavelength
`V_0` = stopping potential
On substituting the respective values in the above formula , we get :
`(6.63 xx 10^-34 xx 3 xx 10^8)/(400 xx 10^-9) = (6.63 xx 10^-34 xx 3 xx 10^8)/λ_0 + 1.6 xx 10^-19 xx 1.1`
`⇒ 4.97 xx 10^-19 = (19.89 xx 10^-26)/λ_0 + 1.76 xx 10^-19`
`⇒ 4.97 = (19.89 xx 10^-7)/λ_0 + 1.76`
`⇒ (19.89 xx 10^-7)/λ_0 = 4.97 - 1.76 = 3.21`
`⇒ λ_0 = (19.89 xx 10^-7)/3.21`
`= 6.196 xx 10^-7 "m" = 620 "nm"`
APPEARS IN
संबंधित प्रश्न
Use the same formula you employ in (a) to obtain electron speed for an collector potential of 10 MV. Do you see what is wrong? In what way is the formula to be modified?
The following graph shows the variation of photocurrent for a photosensitive metal :
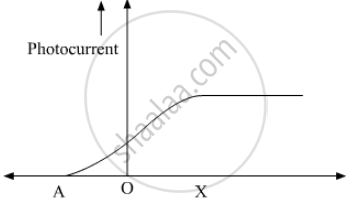
(a) Identify the variable X on the horizontal axis.
(b) What does the point A on the horizontal axis represent?
(c) Draw this graph for three different values of frequencies of incident radiation v1, v2 and v3 (v1 > v2 > v3) for same intensity.
(d) Draw this graph for three different values of intensities of incident radiation I1, I2 and I3 (I1 > I2 > I3) having same frequency.
Can we find the mass of a photon by the definition p = mv?
Is it always true that for two sources of equal intensity, the number of photons emitted in a given time are equal?
If the frequency of light in a photoelectric experiment is doubled, the stopping potential will ______.
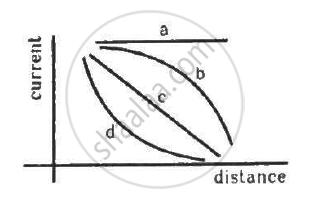
A point source causes photoelectric effect from a small metal plate. Which of the following curves may represent the saturation photocurrent as a function of the distance between the source and the metal?
The work function of a photoelectric material is 4.0 eV. (a) What is the threshold wavelength? (b) Find the wavelength of light for which the stopping potential is 2.5 V.
(Use h = 6.63 × 10-34J-s = 4.14 × 10-15 eV-s, c = 3 × 108 m/s and me = 9.1 × 10-31kg)

On the basis of the graphs shown in the figure, answer the following questions :
(a) Which physical parameter is kept constant for the three curves?
(b) Which is the highest frequency among v1, v2, and v3?
In photoelectric effect, the photoelectric current started to flow. This means that the frequency of incident radiations is ______.
Consider a metal exposed to light of wavelength 600 nm. The maximum energy of the electron doubles when light of wavelength 400 nm is used. Find the work function in eV.
Two monochromatic beams A and B of equal intensity I, hit a screen. The number of photons hitting the screen by beam A is twice that by beam B. Then what inference can you make about their frequencies?
Consider a 20 W bulb emitting light of wavelength 5000 Å and shining on a metal surface kept at a distance 2 m. Assume that the metal surface has work function of 2 eV and that each atom on the metal surface can be treated as a circular disk of radius 1.5 Å.
- Estimate no. of photons emitted by the bulb per second. [Assume no other losses]
- Will there be photoelectric emission?
- How much time would be required by the atomic disk to receive energy equal to work function (2 eV)?
- How many photons would atomic disk receive within time duration calculated in (iii) above?
- Can you explain how photoelectric effect was observed instantaneously?
Why it is the frequency and not the intensity of the light source that determines whether the emission of photoelectrons will occur or not? Explain.
If photons of ultraviolet light of energy 12 eV are incident on a metal surface of work function of 4 eV, then the stopping potential (in eV) will be :
How would the stopping potential for a given photosensitive surface change if the frequency of the incident radiation were increased? Justify your answer.
The figure shows a plot of stopping potential (V0) versus `1/lambda`, where λ is the wavelength of the radiation causing photoelectric emission from a surface. The slope of the line is equal to ______.

Plot a graph showing the variation of photoelectric current, as a function of anode potential for two light beams having the same frequency but different intensities I1 and I2 (I1 > I2). Mention its important features.
What is the effect of threshold frequency and stopping potential on increasing the frequency of the incident beam of light? Justify your answer.
The difference between threshold wavelengths for two metal surfaces A and B having work function ΦA = 9 eV and ΦB = 4.5 eV in nm is ______.
(Given, hc = 1242 eV nm)
