Advertisements
Advertisements
प्रश्न
Using crystal field theory, draw energy level diagram, write electronic configuration of the central metal atom/ion and determine the magnetic moment value in the following:
\[\ce{[CoF6]^{3-}, [Co(H2O)6]^{2+}, [Co(Cn)6]^{3-}}\]
Advertisements
उत्तर
(1) \[\ce{[CoF6]^{3-}}\]: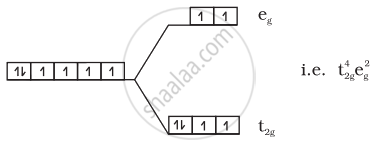
\[\ce{Co^{3+} = 3d^6}\]
Number of unpaired electrons = 4
Magnetic moment = `sqrt(n(n + 2)) = sqrt(4(4 + 2))` = 4.9 BM
(ii) \[\ce{[Co(H2O)6]^{2+}}\]:
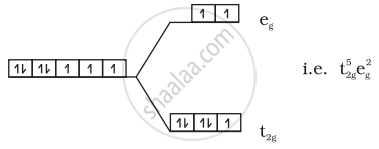
\[\ce{Co^{2+} = 3d^7}\]
Number of unpaired electrons = 3
Magnetic moment = `sqrt(3(3 + 2))` = 3.87 BM
(iii) \[\ce{[Co(CN)6]^{3-}}\]:
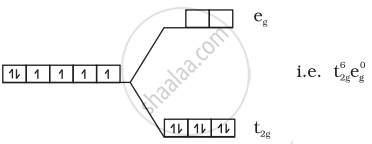
\[\ce{Co^{3+} = 3d^6}\]
No unpaired electrons so diamagnetic.
APPEARS IN
संबंधित प्रश्न
On the basis of crystal field theory, write the electronic configuration for d4 ion if Δ0 > P.
How does the magnitude of Δ0 decide the actual configuration of d orbitals in a coordination entity?
State the superiority of crystal field theory over valence bond theory.
Why are low spin tetrahedral complexes rarely observed?
Write the electronic configuration of Fe(III) on the basis of crystal field theory when it forms an octahedral complex in the presence of (i) strong field, and (ii) weak field ligand. (Atomic no.of Fe=26)
Draw the structures of the following :
(1) XeF6
(2) IF7
Complete and balance the following reactions:
\[\ce{P4 + H2SO4 ->}\] ______ + ______ + ______
The colour of the coordination compounds depends on the crystal field splitting. What will be the correct order of absorption of wavelength of light in the visible region, for the complexes, \[\ce{[Co(NH3)6]^{3+}}\], \[\ce{[Co(CN)6]^{3-}}\], \[\ce{[Co(H2O)6]^{3+}}\]
The CFSE for octahedral \[\ce{[CoCl6]^{4-}}\] is 18,000 cm–1. The CFSE for tetrahedral \[\ce{[CoCl4]^{2-}}\] will be ______.
Atomic number of \[\ce{Mn}\], \[\ce{Fe}\] and \[\ce{Co}\] are 25, 26 and 27 respectively. Which of the following inner orbital octahedral complex ions are diamagnetic?
(i) \[\ce{[Co(NH3)6]^{3+}}\]
(ii) \[\ce{[Mn(CN)6]^{3-}}\]
(iii) \[\ce{[Fe(CN)6]^{4-}}\]
(iv) \[\ce{[Fe(CN)6]^{3-}}\]
An aqueous pink solution of cobalt (II) chloride changes to deep blue on addition of excess of HCl. This is because:
(i) \[\ce{[Co(H2O)6]^{2+}}\] is transformed into \[\ce{[CoCl6]}^{4-}\]
(ii) \[\ce{[Co(H2O)6]^{2+}}\] is transformed into \[\ce{[CoCl4]}^{2-}\]
(iii) tetrahedral complexes have smaller crystal field splitting than octahedral complexes.
(iv) tetrahedral complexes have larger crystal field splitting than octahedral complex.
\[\ce{CuSO4 . 5H2O}\] is blue in colour while \[\ce{CuSO4}\] is colourless. Why?
Match the complex ions given in Column I with the hybridisation and number of unpaired electrons given in Column II and assign the correct code:
| Column I (Complex ion) | Column II (Hybridisation, number of unpaired electrons) |
| A. \[\ce{[Cr(H2O)6]^{3+}}\] | 1. dsp2, 1 |
| B. \[\ce{[Co(CN)4]^{2-}}\] | 2. sp3d2, 5 |
| C. \[\ce{[Ni(NH3)6]^{2+}}\] | 3. d2sp3, 3 |
| D. \[\ce{[MnF6]^{4-}}\] | 4. sp3, 4 |
| 5. sp3d2, 2 |
In a coordination entity, the electronic configuration of the central metal ion is t2g3 eg1
Draw the crystal field splitting diagram for the above complex.
Using crystal field theory, write the electronic configuration of d5 ion, if Δ0 > P.
What is the difference between a weak field ligand and a strong field ligand?
The complex that has highest crystal field splitting energy (Δ) is ______.
Read the passage carefully and answer the questions that follow.
|
Crystal field splitting by various ligands Metal complexes show different colours due to d-d transitions. The complex absorbs light of specific wavelength to promote the electron from t2g to eg level. The colour of the complex is due to the transmitted light, which is complementary of the colour absorbed. The wave number of light absorbed by different complexes of Cr ion are given below:
|
Answer the following questions:
(a) Out of ligands "A", "B", "C" and "D", which ligand causes maximum crystal field splitting? Why?
OR
Which of the two, “A” or “D” will be a weak field ligand? Why?
(b) Which of the complexes will be violet in colour? [CrA6]3- or [CrB6]3+ and why?
(Given: If 560 - 570 nm of light is absorbed, the colour of the complex observed is violet.)
(c) If the ligands attached to Cr3+ ion in the complexes given in the table above are water, cyanide ion, chloride ion, and ammonia (not in this order).
Identify the ligand, write the formula and IUPAC name of the following:
- [CrA6]3-
- [CrC6]3+
The value of the spin only magnetic moment for one of the following configurations is 2.84 BM. The correct one is:
