Advertisements
Advertisements
प्रश्न
Solve the numerical problem.
Calculate the wavelength in nm of an X-ray wave of frequency 2.0 × 1018 Hz.
Advertisements
उत्तर
Given: c = 3 × 108 , ν = 2 × 1018 Hz
To find: Wavelength (λ)
Formula: c = νλ
Calculation: From formula,
λ = `"c"/ν=(3xx10^8)/(2xx10^18)` = 1.5 × 10−10 = 0.15 nm
The wavelength of an X-ray is 0.15 nm.
APPEARS IN
संबंधित प्रश्न
What role dose infra-red radiation play in physical therapy?
To which part of the electromagnetic spectrum does a wave of frequency 5 × 1011 Hz belong?
A radio can tune in to any station in the 7.5 MHz to 12 MHz band. What is the corresponding wavelength band?
State an additional use of the X-rays waves
What is the range of the wavelength of the following electromagnetic waves?
(a) Visible.
What is the range of the wavelength of the following electromagnetic waves?
(a) Infrared.
Name the rays or waves of highest frequency .
Two waves A and B have wavelength 0.01 Å and 9000 Å respectively.
- Name the two waves.
- Compare the speeds of these waves when they travel in vacuum.
Name three properties of ultraviolet radiations which are similar to visible light.
Can X-rays be used for photoelectric effect?
If the current in the circuit for heating the filament is increased, the cutoff wavelength
The figure shows the intensity-wavelength relations of X-rays coming from two different Coolidge tubes. The solid curve represents the relation for the tube A in which the potential difference between the target and the filament is VA and the atomic number of the target material is ZA. These quantities are VB and ZB for the other tube. Then,

For harder X-rays,
(a) the wavelength is higher
(b) the intensity is higher
(c) the frequency is higher
(d) the photon energy is higher.
The Kβ X-rays from certain elements are given below. Draw a Moseley-type plot of √v versus Z for Kβ radiation.
| Element | Ne | P | Ca | Mn | Zn | Br |
| Energy (keV) | 0.858 | 2.14 | 4.02 | 6.51 | 9.57 | 13.3 |
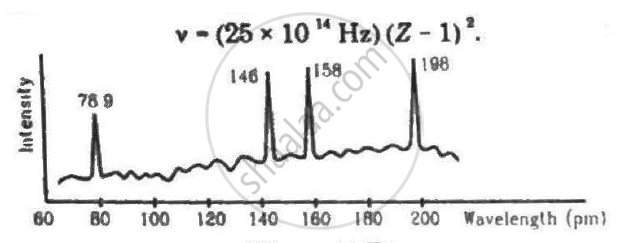
Continuous X-rays are made to strike a tissue paper soaked with polluted water. The incoming X-rays excite the atoms of the sample by knocking out the electrons from the inner shells. Characteristic X-rays are analysed and the intensity is plotted against the wavelength. Assuming that only Kα intensities are detected, list the elements present in the sample from the plot. Use Moseley's equation v − (25 × 1014Hz)(Z − 1)2.
(Use Planck constant h = 6.63 × 10-34 Js= 4.14 × 10-15 eVs, speed of light c = 3 × 108 m/s.)
To which regions of the electromagnetic spectrum do the following wavelengths belong:
(a) 250 nm
(b) 1500 nm
Name the part of the electromagnetic spectrum which is:
Produced by bombarding a metal target with high electrons.
Identify the part of the electromagnetic spectrum used in (i) radar and (ii) eye surgery. Write their frequency range.
Give one use of electromagnetic radiation in Ultraviolet radiation.
Answer briefly.
Can we produce a pure electric or magnetic wave in space? Why?
Answer briefly.
Name the most harmful radiation entering the Earth's atmosphere from outer space.
Radio waves of constant amplitude can be generated with.
Electromagnetic waves with wavelength
- λ1 is used in satellite communication.
- λ2 is used to kill germs in water purifies.
- λ3 is used to detect leakage of oil in underground pipelines.
- λ4 is used to improve visibility in runways during fog and mist conditions.
- Identify and name the part of electromagnetic spectrum to which these radiations belong.
- Arrange these wavelengths in ascending order of their magnitude.
- Write one more application of each.
Electromagnetic waves of wavelengths λ1, λ2 and λ3 are used in a radar system, in water purifiers and in remote switches of TV, respectively.
- Identify the electromagnetic waves.
- Write one source for each of them.
Identify the electromagnetic wave whose wavelength range is from about 10-12 m to about 10-8 m. Write one use of this.
Identify the electromagnetic wave whose wavelength range is from about 10-3 m to about 10-1 m. Write one use of this.
Identify the part of the electromagnetic spectrum which:
- produces the heating effect.
- is absorbed by the ozone layer in the atmosphere.
- is used for studying crystal structure.
Write any one method of the production of each of the above radiations.
Assertion (A): Ultraviolet radiations is scattered more as compared to the microwave radiations.
Reason (R): Wavelength of ultraviolet radiation is more than the wavelength of microwave radiation.
Name the electromagnetic radiation whose frequency is 10 Hz.
Name one radiation having the wavelength longer than the wavelength of these radiations.
