Advertisements
Advertisements
प्रश्न
What role dose infra-red radiation play in maintain the Earth’s warmth
Advertisements
उत्तर
Visible light from the atmosphere is absorbed by the Earth’s surface and radiated as infrared radiation which gets trapped by greenhouse gases such as carbon dioxide and water vapour. This is how infrared radiation also plays an important role in maintaining the Earth’s warmth
APPEARS IN
संबंधित प्रश्न
Why are microwaves considered suitable for radar systems used in aircraft navigation?
Identify the part of the electromagnetic spectrum which is:
produced by bombarding a metal target by high speed electrons.
State the approximate range of wavelength associated with the ultraviolet rays.
50% of the X-ray coming from a Coolidge tube is able to pass through a 0.1 mm thick aluminium foil. The potential difference between the target and the filament is increased. The thickness of the aluminium foil that will allow 50% of the X-ray to pass through will be
The X-ray coming from a Coolidge tube has a cutoff wavelength of 80 pm. Find the kinetic energy of the electrons hitting the target.
(Use Planck constant h = 6.63 × 10-34 Js= 4.14 × 10-15 eVs, speed of light c = 3 × 108 m/s.)
Heat at the rate of 200 W is produced in an X-ray tube operating at 20 kV. Find the current in the circuit. Assume that only a small fraction of the kinetic energy of electrons is converted into X-rays.
For television broadcasting, the frequency employed is normally
Identify the electromagnetic wave whose wavelength range is from about 10-3 m to about 10-1 m. Write one use of this.
Name the electromagnetic radiation that has been used in obtaining the image below.
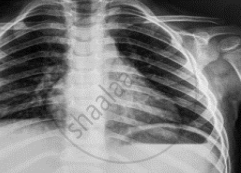
What is the wavelength range of electromagnetic radiation used in radio broadcast?
