Advertisements
Advertisements
प्रश्न
State the approximate range of wavelength associated with the ultraviolet rays.
Advertisements
उत्तर
Ultraviolet rays wavelength range 100 Å to 4000 Å.
APPEARS IN
संबंधित प्रश्न
Name the high energetic invisible electromagnetic waves which help in the study of the structure of crystals
Name the waves of wavelength nearly 0.1 nm.
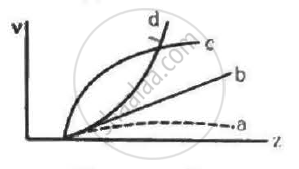
Frequencies of Kα X-rays of different materials are measured. Which one of the graphs in the figure may represent the relation between the frequency v and the atomic number Z ?
What potential difference should be applied across an X-ray tube to get X-ray of wavelength not less than 0.10 nm? What is the maximum energy of a photon of this X-ray in joule?
(Use Planck constant h = 6.63 × 10-34 Js= 4.14 × 10-15 eVs, speed of light c = 3 × 108 m/s.)
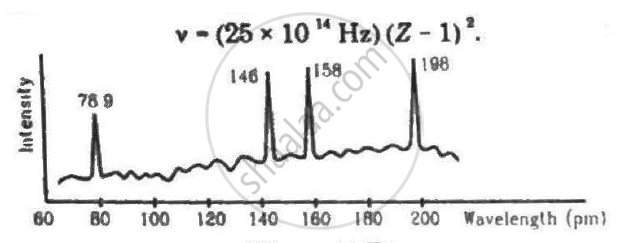
Continuous X-rays are made to strike a tissue paper soaked with polluted water. The incoming X-rays excite the atoms of the sample by knocking out the electrons from the inner shells. Characteristic X-rays are analysed and the intensity is plotted against the wavelength. Assuming that only Kα intensities are detected, list the elements present in the sample from the plot. Use Moseley's equation v − (25 × 1014Hz)(Z − 1)2.
(Use Planck constant h = 6.63 × 10-34 Js= 4.14 × 10-15 eVs, speed of light c = 3 × 108 m/s.)
Name three radiations and their wavelength range which are invisible and beyond the violet end of the visible spectrum.
Name the radiation of the electromagnetic spectrum which is used for the following:
Radar and Give the frequency range.
Give one use of electromagnetic radiation in Ultraviolet radiation.
Choose the correct option.
Earth’s atmosphere is richest in
The half-value thickness of an absorber is defined as the thickness that will reduce exponentially the intensity of a beam of particles by a factor of 2. The half-value thickness in (µm) for lead assuming X-ray beam of wavelength 20 pm, µ = 50 cm-1 for X-rays in lead at wavelength λ = 20 pm, is ______ µm.
