Advertisements
Advertisements
प्रश्न
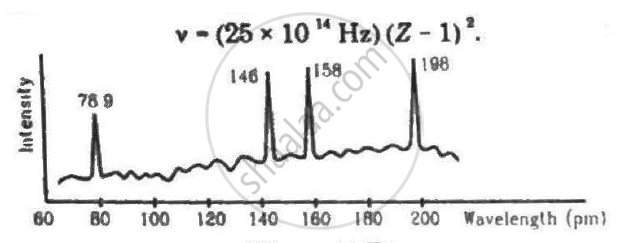
Continuous X-rays are made to strike a tissue paper soaked with polluted water. The incoming X-rays excite the atoms of the sample by knocking out the electrons from the inner shells. Characteristic X-rays are analysed and the intensity is plotted against the wavelength. Assuming that only Kα intensities are detected, list the elements present in the sample from the plot. Use Moseley's equation v − (25 × 1014Hz)(Z − 1)2.
(Use Planck constant h = 6.63 × 10-34 Js= 4.14 × 10-15 eVs, speed of light c = 3 × 108 m/s.)
Advertisements
उत्तर
Given :-
`f = (25 × 10^14 "Hz") (Z − 1)^2`
`or c/lambda = 25 xx 10^14(Z - 1)^2`
(a) `(3 xx 10^8)/(78.9 xx 10^-12 xx 25 xx 10^14)` = `(Z - 1)^2`
or `(Z - 1)^2 = 0.001520 xx 10^6 = 1520`
`⇒ Z - 1 = 38.98`
`or Z = 39.98 = 40`
Thus , it is (Zr) .
(b) `(3 xx 10^8)/(146 xx 10^-12 xx 25 xx 10^14)` = `(Z - 1)^2`
`or (Z - 1)^2 = 0.00082219 xx 10^6`
`or Z - 1 = 28.669`
`or Z = 29.669 = 30`
Thus , it is (Zn).
(c) `(3 xx 10^8)/(158 xx 10^-12 xx 25 xx 10^14) = (Z - 1)^2`
`or (Z -1)^2 = 0.0007594 xx 10^4`
`or Z - 1 = 27.5559`
`or Z - 1 = 28.5589 = 29`
Thus , it is (Cu).
(d) `(3 xx 10^8)/(198 xx 10^-12 xx 25 xx 10^14) = (Z - 1)^2`
`or (Z - 1)^2 = 0.000606 xx 10^4`
`or Z-1 = 24.6162`
`or Z = 25.6162 = 26`
Thus , it is (Fe).
APPEARS IN
संबंधित प्रश्न
Identify the part of the electromagnetic spectrum which is:
produced by bombarding a metal target by high speed electrons.
A radio can tune in to any station in the 7.5 MHz to 12 MHz band. What is the corresponding wavelength band?
Given below are some famous numbers associated with electromagnetic radiations in different contexts in physics. State the part of the electromagnetic spectrum to which each belongs.
(a) 21 cm (wavelength emitted by atomic hydrogen in interstellar space).
(b) 1057 MHz (frequency of radiation arising from two close energy levels in hydrogen; known as Lamb shift).
(c) 2.7 K [temperature associated with the isotropic radiation filling all space-thought to be a relic of the ‘big-bang’ origin of the universe].
(d) 5890 Å - 5896 Å [double lines of sodium]
(e) 14.4 keV [energy of a particular transition in 57Fe nucleus associated with a famous high resolution spectroscopic method (Mössbauer spectroscopy)].
The small ozone layer on top of the stratosphere is crucial for human survival. Why?
Which part of electromagnetic spectrum is used in radar systems?
Cutoff wavelength of X-rays coming from a Coolidge tube depends on the
(a) target material
(b) accelerating voltage
(c) separation between the target and the filament
(d) temperature of the filament.
The electron beam in a colour TV is accelerated through 32 kV and then strikes the screen. What is the wavelength of the most energetic X-ray photon?
(Use Planck constant h = 6.63 × 10-34 Js= 4.14 × 10-15 eVs, speed of light c = 3 × 108 m/s.)
Name three radiations and their wavelength range which are invisible and beyond the violet end of the visible spectrum.
Name two sources of infrared radiation.
Answer briefly.
Why are microwaves used in radar?
Answer briefly.
Give two uses of ultraviolet rays.
Answer briefly.
What are radio waves?
If the Earth did not have atmosphere, would its average surface temperature be higher or lower than what it is now? Explain.
Electromagnetic waves of wavelengths λ1, λ2 and λ3 are used in a radar system, in water purifiers and in remote switches of TV, respectively.
- Identify the electromagnetic waves.
- Write one source for each of them.
In uranium (Z = 92) the K absorption edge is 0.107 Å and the Kα line is 0.126 Å, and the wavelength of the L absorption edge is ______.
Which is the correct ascending order of wavelengths?
Assertion (A): Ultraviolet radiations is scattered more as compared to the microwave radiations.
Reason (R): Wavelength of ultraviolet radiation is more than the wavelength of microwave radiation.
In an atom X, electrons absorb the energy from an external source. This energy “excites” the electrons from a lower-energy level to a higher-energy level around the nucleus of the atom. When electrons return to the ground state, they emit photons.
The figure below is the energy level diagram of atom X with three energy levels, E1 = 0.00eV, E2 = 1.78eV and E3 = 2.95eV. The ground state is considered 0 eV for reference. The transition of electrons takes place between levels E1 and E2.

- What wavelength of radiation is needed to excite the atom to energy level E2 from E1?
- Suppose the external source has a power of 100 W. What would be the rate of photon emission?
