Advertisements
Advertisements
प्रश्न
One requires 11eV of energy to dissociate a carbon monoxide molecule into carbon and oxygen atoms. The minimum frequency of the appropriate electromagnetic radiation to achieve the dissociation lies in ______.
पर्याय
visible region.
infrared region.
ultraviolet region.
microwave region.
Advertisements
उत्तर
One requires 11eV of energy to dissociate a carbon monoxide molecule into carbon and oxygen atoms. The minimum frequency of the appropriate electromagnetic radiation to achieve the dissociation lies in ultraviolet region.
Explanation:
Given energy required to dissociate a carbon monoxide moleule into carbon and oxygen atoms E = 11eV
We know that E = hv, where h = 6.62 × 10–34 J – s
v = frequency
⇒ 11 eV = hv
⇒ ` v = (11 xx 1.6 xx 10^-19)/(6.62 xx 10^-34) J`
= `2.65 xx 10^15` Hz
This frequency radiation belongs to ultraviolet region.
APPEARS IN
संबंधित प्रश्न
To which part of electromagnetic spectrum does a wave of frequency 3 × 1013 Hz belong?
A wave has wavelength 50 Å.
- Name the wave.
- State its speed in vacuum.
- State its one use.
Which radiation is used for satellite communication?
What is the range of the wavelength of the following electromagnetic waves?
(a) Gamma rays.
Name the region beyond the red end of the spectrum.
State the approximate range of wavelength associated with the ultraviolet rays.
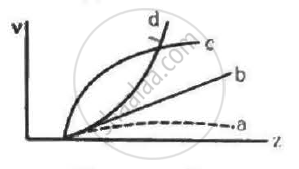
Frequencies of Kα X-rays of different materials are measured. Which one of the graphs in the figure may represent the relation between the frequency v and the atomic number Z ?
The X-ray beam emerging from an X-ray tube
Find the cutoff wavelength for the continuous X-rays coming from an X-ray tube operating at 30 kV.
(Use Planck constant h = 4.14 × 10-15 eVs, speed of light c = 3 × 108 m/s.)
If the operating potential in an X-ray tube is increased by 1%, by what percentage does the cutoff wavelength decrease?
(Use Planck constant h = 6.63 × 10-34 Js= 4.14 × 10-15 eVs, speed of light c = 3 × 108 m/s.)
The short-wavelength limit shifts by 26 pm when the operating voltage in an X-ray tube is increased to 1.5 times the original value. What was the original value of the operating voltage?
(Use Planck constant h = 6.63 × 10-34 Js= 4.14 × 10-15 eVs, speed of light c = 3 × 108 m/s.)
Name the scientist who discovered Infra-red waves
State the name and the range of wavelength of the invisible electromagnetic waves beyond the red end of the visible spectrum.
Calculate the shortest wavelength of electromagnetic radiation present in Balmer series of hydrogen spectrum.
How will you investigate the existence of the radiation beyond the red and violet extremes of the spectrum?
Give one use of electromagnetic radiation in Microwaves.
State two uses of infrared radiations.
Identify the electromagnetic wave whose wavelength range is from about 10-12 m to about 10-8 m. Write one use of this.
