Advertisements
Advertisements
Question
One requires 11eV of energy to dissociate a carbon monoxide molecule into carbon and oxygen atoms. The minimum frequency of the appropriate electromagnetic radiation to achieve the dissociation lies in ______.
Options
visible region.
infrared region.
ultraviolet region.
microwave region.
Advertisements
Solution
One requires 11eV of energy to dissociate a carbon monoxide molecule into carbon and oxygen atoms. The minimum frequency of the appropriate electromagnetic radiation to achieve the dissociation lies in ultraviolet region.
Explanation:
Given energy required to dissociate a carbon monoxide moleule into carbon and oxygen atoms E = 11eV
We know that E = hv, where h = 6.62 × 10–34 J – s
v = frequency
⇒ 11 eV = hv
⇒ ` v = (11 xx 1.6 xx 10^-19)/(6.62 xx 10^-34) J`
= `2.65 xx 10^15` Hz
This frequency radiation belongs to ultraviolet region.
APPEARS IN
RELATED QUESTIONS
Name the subjective property of light related to its wavelength.
What is the range of the wavelength of the following electromagnetic waves?
(A) Radio waves.
Name the region beyond the red end of the spectrum.
Name of physical quantity which remains same for microwaves of wavelength 1 mm and UV radiations of 1600 Å in vacuum.
Can a hydrogen atom emit characteristic X-rays?
Why is exposure to X-rays injurious to health but not exposure to visible light, when both are electromagnetic waves?
For harder X-rays,
(a) the wavelength is higher
(b) the intensity is higher
(c) the frequency is higher
(d) the photon energy is higher.
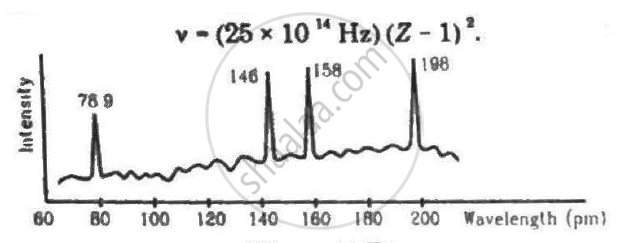
Continuous X-rays are made to strike a tissue paper soaked with polluted water. The incoming X-rays excite the atoms of the sample by knocking out the electrons from the inner shells. Characteristic X-rays are analysed and the intensity is plotted against the wavelength. Assuming that only Kα intensities are detected, list the elements present in the sample from the plot. Use Moseley's equation v − (25 × 1014Hz)(Z − 1)2.
(Use Planck constant h = 6.63 × 10-34 Js= 4.14 × 10-15 eVs, speed of light c = 3 × 108 m/s.)
Name the scientist who discovered radio waves
Name the scientist who discovered Ultraviolet rays
Define the term "Intensity" in the photon picture of electromagnetic radiation.
Give one use of electromagnetic radiations in Infrared radiation.
Answer briefly.
Name the most harmful radiation entering the Earth's atmosphere from outer space.
Why does microwave oven heats up a food item containing water molecules most efficiently?
The magnetic field of a beam emerging from a filter facing a floodlight is given by B0 = 12 × 10–8 sin (1.20 × 107z – 3.60 × 1015t) T. What is the average intensity of the beam?
What happens to the intensity of light from a bulb if the distance from the bulb is doubled? As a laser beam travels across the length of a room, its intensity essentially remains constant. What geometrical characteristic of LASER beam is responsible for the constant intensity which is missing in the case of light from the bulb?
Write two uses of the following radiation.
Gamma rays
What is the speed of radio waves in vacuum?
Name one radiation having the wavelength longer than the wavelength of these radiations.
Name the electromagnetic radiation that has been used in obtaining the image below.