Advertisements
Advertisements
प्रश्न
Explain primary benzylic halide shows higher reactivity by SN1 mechanism than other primary alkyl halide.
Advertisements
उत्तर
- SN1 reaction involves the formation of a carbocation intermediate. The benzylic carbocation intermediate formed are resonance stabilized, and hence SN1 mechanism is favoured.
- Resonance stabilization of benzylic carbocation can be represented as:

- Reactivity order of alkyl halides towards SN1 mechanism is 3° > 2° > 1° > CH3X. That is, SN1 mechanism is least favoured in primary alkyl halides.
- Benzylic carbocation formed is more stable than the primary carbocation; therefore, primary benzylic halide shows higher reactivity by SN1 mechanism than other primary alkyl halide.
APPEARS IN
संबंधित प्रश्न
from the following pair would undergo SN2 faster from the other?
Arrange the following in the increasing order of boiling points.
- 1-Bromopropane
- 2- Bromopropane
- 1- Bromobutane
- 1-Bromo-2-methylpropane
Give reasons:
Haloarenes are less reactive than haloalkanes.
Convert the following:
Propene to propan-1-ol
Convert the following:
Ethanol to propane nitrile
Complete the following reaction sequence by writing the structural formulae of the organic compound 'A', 'B' and 'C'.
\[\ce{2-Bromobutane->[Alc.KOH]A->[][Br2]B->[][NaNH2]C}\]
Complete the following reaction sequence by writing the structural formulae of the organic compound 'A', 'B' and 'C'.
\[\ce{Isopropyl alcohol ->[\triangle][PBr3] A ->[][NH3 excess] B}\]
Observe the following and answer the question given below.

Can it react by SN1 mechanism? Justify your answer.
Nucleophilic substitution reaction of 2, 4-dinitrochlorobenzene is faster than p-nitrochlorobenzene. Give reason.
Which of the following is a primary halide?
The least reactive towards nucleophilic addition reactions is ____________.
Which of the following is least reactive towards SN1 reactions?
Dehydrohalogenation of an alkyl halide is ____________.
\[\ce{C6H5CH2Cl + KCN(alc) -> X + Y}\]
Compounds X and Y are ____________.
When C2H5Br is treated with excess of alc. NH3, the major product obtained is ____________.
What is the major product obtained in the sulphonation of chlorobenzene with concentrated sulphuric acid?
Explain the dehydrohalogenation reaction of 2-chlorobutane.
Complete the following reactions giving a major product.
\[\begin{array}{cc}
\ce{CH3}\phantom{................}\\
|\phantom{..................}\\
\ce{CH3 - C - CH2 - Cl ->[Na/dry ether] A}\\
|\phantom{..................}\\
\ce{CH3}\phantom{................}
\end{array}\]
Complete the following reaction giving a major product.
\[\begin{array}{cc}
\ce{CH3}\phantom{.................}\\
|\phantom{...................}\\
\ce{CH3 - C - CH2 - Cl->[Na/dry ether]A}\\
|\phantom{..................}\\
\ce{CH3}\phantom{................}
\end{array}\]
Observe the following and answer the question given below:
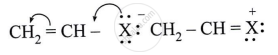
Comment on the bond length of C-X bond in it.
Observe the following and answer the question given below:
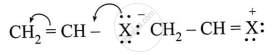
Can react by SN1 mechanism? Justify your answer.
Complete the following reactions giving major product.
\[\begin{array}{cc}
\ce{CH3}\phantom{................}\\
|\phantom{...................}\\
\ce{CH3 - C - CH2 - Cl ->[Na/dry ether] A}\\
|\phantom{...................}\\
\ce{CH3}\phantom{.................}
\end{array}\]
Complete the following reaction giving major product.
\[\begin{array}{cc}
\ce{CH3}\phantom{...............}\\
|\phantom{..................}\\
\ce{CH3 - C - CH2 - Cl->[Na/dry ether]A}\\
|\phantom{..................}\\
\ce{CH3}\phantom{...............}\\
\end{array}\]
What is the action of following on ethyl bromide?
alcoholic sodium hydroxide
Complete the following reaction giving major product.
\[\begin{array}{cc}
\ce{CH3}\phantom{...............}\\
|\phantom{..................}\\
\ce{CH3 - C - CH2 - Cl ->[Na/dry ether]A}\\
|\phantom{...................}\\
\ce{CH3}\phantom{.................}
\end{array}\]
Complete the following reaction giving major product.
\[\begin{array}{cc}
\ce{CH3}\phantom{.................}\\
|\phantom{...................}\\
\ce{CH3 - C - CH2 - Cl ->[Na/dry ether] A}\\
|\phantom{...................}\\
\ce{CH3}\phantom{.................}
\end{array}\]
Complete the following reaction giving major product.
\[\begin{array}{cc}
\ce{CH3}\phantom{................}\\
|\phantom{...................}\\
\ce{CH3 - C - CH2 - Cl ->[Na/dry ether] A}\\
|\phantom{...................}\\
\ce{CH3}\phantom{................}\\
\end{array}\]
Identify the substrate 'X' in the following reaction.
\[\ce{\underset{}{X + O2} ->[i) Co-naphtenate 423 K][ii) dil HClΔ] \underset{\underset{}{}}{Phenol + Acetone}}\]
Which among the following is NOT benzylic halide?
