Advertisements
Advertisements
प्रश्न
Nucleophilic substitution reaction of 2, 4-dinitrochlorobenzene is faster than p-nitrochlorobenzene. Give reason.
Advertisements
उत्तर
- The presence of electron-withdrawing (–NO2) group at ortho and para position greatly increases the reactivity of 2, 4-dinitrochlorobenzene towards substitution of halogen atom.
- Greater the number of electron-withdrawing (–NO2) groups at o and p position, greater is the reactivity. Therefore, nucleophilic substitution reaction of 2, 4-dinitrochlorobenzene is faster than p-nitrochlorobenzene.
APPEARS IN
संबंधित प्रश्न
from the following pair would undergo SN2 faster from the other?
Complete the following reaction giving major product.
\[\begin{array}{cc}\ce{CH3\phantom{................}}\\
|\phantom{...................}\\
\ce{CH3 - C - CH2 - Cl ->[Na/dry ether] A}\\
|\phantom{...................}\\
\ce{CH3\phantom{................}}
\end{array}\]
Name the reagent used to bring about the following conversion.
Chlorobenzene to biphenyl
Give reasons:
Haloarenes are less reactive than haloalkanes.
Convert the following:
Propene to propan-1-ol
Convert the following:
Ethanol to propane nitrile
HCl is added to a hydrocarbon ‘A’ \[\ce{(C4H8)}\] to give a compound ‘B’ which on hydrolysis with aqueous alkali forms tertiary alcohol ‘C’ \[\ce{(C4H10O)}\]. Identify ‘A’ ,‘B’ and ‘C’.
Complete the following reaction sequence by writing the structural formulae of the organic compound 'A', 'B' and 'C'.
\[\ce{2-Bromobutane->[Alc.KOH]A->[][Br2]B->[][NaNH2]C}\]
Complete the following reaction sequence by writing the structural formulae of the organic compound 'A', 'B' and 'C'.
\[\ce{Isopropyl alcohol ->[\triangle][PBr3] A ->[][NH3 excess] B}\]
Observe the following and answer the question given below.

Name the type of halogen derivative.
Observe the following and answer the question given below.

Can it react by SN1 mechanism? Justify your answer.
Explain. Aryl halides are less reactive than alkyl halides towards nucleophilic substitution reactions.
Which of the following is least reactive towards SN1 reactions?
Ethyl bromide undergoes the following reaction:
\[\ce{\underset{Ethyl bromide}{C2H5Br} + \underset{(aq.)}{KOH} ->[\Delta] \underset{Ethyl alcohol}{C2H5OH} + KBr}\]
Which of the following is a WRONG statement?
How many metameric ethers are represented by the molecular formula C4H10O?
What type of hybridisation is present in carbocation formed during the alkaline hydrolysis of 1- bromo-1- phenyl ethane?
Which of the following is FALSE regarding SN2 reaction mechanism?
The SN2 reaction of a compound containing an asymmetric carbon atom always gives ____________.
The major product formed when 2-bromobutane is treated with alcoholic KOH is ______.
The order of reactivity of the given haloalkanes towards nucleophiles is:
With which halogen the reactions, of alkanes are explosive?
The compound that reacts the fastest with sodium methoxide is ______.
Observe the following and answer the questions given below:
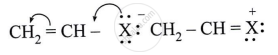
Name the type of halogen derivative.
Observe the following and answer the question given below:
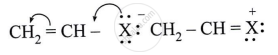
Comment on the bond length of C-X bond in it.
Observe the following and answer the question given below:
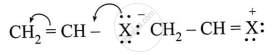
Can react by SN1 mechanism? Justify your answer.
Define and explain the SN1 mechanism with a suitable example.
Identify the chiral molecule from the following.
Give a reason for the following:
Ethoxy ethane with conc. HI at 373 K gives C2H5OH and CH3I but not CH3OH and C2H5I.
