Advertisements
Advertisements
प्रश्न
Write the product formed when alkyl halide reacts with silver nitrite.
Advertisements
उत्तर
\[\ce{\underset{(Alkyl halide)}{R - X} + \underset{(Silver nitrite)}{Ag NO2} -> \underset{Nitroalkane}{R - NO2 + Ag - X}}\]
Nitroalkane is formed when alkyl halide reacts with silver nitrite.
APPEARS IN
संबंधित प्रश्न
Complete the following reaction giving major products.

Convert the following:
Propene to propan-1-ol
Convert the following:
Benzyl alcohol to benzyl cyanide
Observe the following and answer the question given below.

Can it react by SN1 mechanism? Justify your answer.
Write the correct order of increasing ease of dehydrohalogenation.
\[\ce{\underset{\text{(I)}}{CH3 - CH2 - CH2 - Cl}}\]
\[\ce{\underset{\text{(II)}}{CH3 - CH(Cl) - CH3}}\]
\[\begin{array}{cc}
\ce{CH3}\\
|\\
\ce{CH3-C-Cl}\phantom{...}\\
|\\
\ce{CH3}\\
\ce{(III)}
\end{array}\]
Explain primary benzylic halide shows higher reactivity by SN1 mechanism than other primary alkyl halide.
Explain the factors affecting SN1 and SN2 mechanism.
What is dehydrohalogenation? State the rule for the formation of the preferred product of dehydrohalogenation.
The least reactive towards nucleophilic addition reactions is ____________.
The following mechanism has been proposed for the reaction of NO2 with F2 to form NO2F:
\[\ce{NO2_{(g)} + F2_{(g)} -> NO2F_{(g)} + F_{(g)} (slow)}\]
\[\ce{F_{(g)} + NO2_{(g)} -> NO2F_{(g)} (fast)}\]
The order of the reaction with respect to NO2(g)
What type of hybridisation is present in carbocation formed during the alkaline hydrolysis of 1- bromo-1- phenyl ethane?
When C2H5Br is treated with excess of alc. NH3, the major product obtained is ____________.
Convert the following.
p-Nitrochlorobenzene to p-nitrophenol
The compound that reacts the fastest with sodium methoxide is ______.
Explain the dehydrohalogenation reaction of 2-chlorobutane.
Observe the following and answer the questions given below:
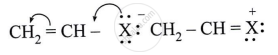
Name the type of halogen derivative.
Observe the following and answer the question given below:
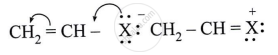
Comment on the bond length of C-X bond in it.
Discuss β - elimination reaction.
Complete the following reactions giving major product.
\[\begin{array}{cc}
\ce{CH3}\phantom{................}\\
|\phantom{...................}\\
\ce{CH3 - C - CH2 - Cl ->[Na/dry ether] A}\\
|\phantom{...................}\\
\ce{CH3}\phantom{.................}
\end{array}\]
Define and explain the SN1 mechanism with a suitable example.
Complete the following reaction giving major product.
\[\begin{array}{cc}
\ce{CH3}\phantom{...............}\\
|\phantom{..................}\\
\ce{CH3 - C - CH2 - Cl->[Na/dry ether]A}\\
|\phantom{..................}\\
\ce{CH3}\phantom{...............}\\
\end{array}\]
Identify the chiral molecule from the following.
Complete the following reaction giving major product.
\[\begin{array}{cc}
\ce{CH3}\phantom{...............}\\
|\phantom{..................}\\
\ce{CH3 - C - CH2 - Cl ->[Na/dry ether]A}\\
|\phantom{...................}\\
\ce{CH3}\phantom{.................}
\end{array}\]
Complete the following reaction giving major product.
\[\begin{array}{cc}
\ce{CH3}\phantom{................}\\
|\phantom{..................}\\
\ce{CH3 - C - CH2 - Cl ->[Na/dry ether]A}\\
|\phantom{..................}\\
\ce{CH3}\phantom{................}
\end{array}\]
Complete the following reaction giving major product.
\[\begin{array}{cc}
\ce{CH3}\phantom{.................}\\
|\phantom{...................}\\
\ce{CH3 - C - CH2 - Cl ->[Na/dry ether] A}\\
|\phantom{...................}\\
\ce{CH3}\phantom{.................}
\end{array}\]
Complete the following reaction giving major product.
\[\begin{array}{cc}
\ce{CH3}\phantom{................}\\
|\phantom{...................}\\
\ce{CH3 - C - CH2 - Cl ->[Na/dry ether] A}\\
|\phantom{...................}\\
\ce{CH3}\phantom{................}\\
\end{array}\]
Which of the following pair of compounds on heating gives butanenitrile?
