Advertisements
Advertisements
प्रश्न
Explain the dehydrohalogenation reaction of 2-chlorobutane.
Advertisements
उत्तर
When 2-chlorobutane is boiled with an alcoholic solution of potassium hydroxide, it undergoes the elimination of the hydrogen atoms from β-carbon and chlorine atoms from α-carbon resulting in the formation of But-2-ene and But-1-ene.
Reaction:

But according to Saytzeff's rule, in dehydrohalogenation reaction, the preferred product is that alkene which has a greater number of alkyl groups attached to doubly bonded carbon atoms.
Hence, But-2-ene is obtained as the major product in the dehydrohalogenation reaction of 2-chlorobutane.
APPEARS IN
संबंधित प्रश्न
Complete the following reaction giving major products.
\[\ce{CH3 - CH = CH2 ->[HBr][peroxide] A ->[alc. KOH] B}\]
Name the reagent used to bring about the following conversion.
Ethyl bromide to ethyl isocyanide
Arrange the following in the increasing order of boiling points.
- 1-Bromopropane
- 2- Bromopropane
- 1- Bromobutane
- 1-Bromo-2-methylpropane
Distinguish between SN1 and SN2 mechanism of substitution reaction.
Convert the following:
Benzyl alcohol to benzyl cyanide
Convert the following:
Ethanol to propane nitrile
HCl is added to a hydrocarbon ‘A’ \[\ce{(C4H8)}\] to give a compound ‘B’ which on hydrolysis with aqueous alkali forms tertiary alcohol ‘C’ \[\ce{(C4H10O)}\]. Identify ‘A’ ,‘B’ and ‘C’.
Complete the following reaction sequence by writing the structural formulae of the organic compound 'A', 'B' and 'C'.
\[\ce{2-Bromobutane->[Alc.KOH]A->[][Br2]B->[][NaNH2]C}\]
Observe the following and answer the question given below.

Comment on the bond length of C–X bond in it.
Explain. Aryl halides are less reactive than alkyl halides towards nucleophilic substitution reactions.
Explain primary benzylic halide shows higher reactivity by SN1 mechanism than other primary alkyl halide.
Explain aqueous alkaline hydrolysis of tert. butyl bromide.
In alkaline hydrolysis of tertiary butyl bromide, ____________.
The least reactive towards nucleophilic addition reactions is ____________.
Ethyl bromide undergoes the following reaction:
\[\ce{\underset{Ethyl bromide}{C2H5Br} + \underset{(aq.)}{KOH} ->[\Delta] \underset{Ethyl alcohol}{C2H5OH} + KBr}\]
Which of the following is a WRONG statement?
Nitroalkanes are obtained in laboratory from primary or secondary alkyl halides by the action of ______.
How many metameric ethers are represented by the molecular formula C4H10O?
Identify major product 'B' in the following reaction.
\[\ce{But-1-ene ->[HBr][Peroxide] A ->[AgCN][\Delta] B}\]
Which of the following statements is incorrect regarding the dehydrohalogenation of alkenes?
Complete the following reactions giving a major product.
\[\begin{array}{cc}
\ce{CH3}\phantom{................}\\
|\phantom{..................}\\
\ce{CH3 - C - CH2 - Cl ->[Na/dry ether] A}\\
|\phantom{..................}\\
\ce{CH3}\phantom{................}
\end{array}\]
Observe the following and answer the questions given below:
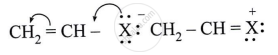
Name the type of halogen derivative.
Discuss β - elimination reaction.
Complete the following reaction sequences by writing the structural formulae of the organic compounds 'A', 'B' and 'C'.
\[\ce{2-Bromobutan ->[alc. KOH] A ->[][Br2] B ->[][NANH2] C}\]
Define and explain the SN1 mechanism with a suitable example.
Complete the following reaction giving major product.
\[\begin{array}{cc}
\ce{CH3}\phantom{.................}\\
|\phantom{...................}\\
\ce{CH3 - C - CH2 - Cl ->[Na/dry ether] A}\\
|\phantom{...................}\\
\ce{CH3}\phantom{.................}\\
\end{array}\]
