Advertisements
Advertisements
प्रश्न
Explain aqueous alkaline hydrolysis of tert. butyl bromide.
Advertisements
उत्तर
i. Aqueous alkaline hydrolysis of tert. butyl bromide forms tert-butyl alcohol. The reaction can be given as,
\[\begin{array}{cc}
\ce{\phantom{....}CH3\phantom{.........................}}\ce{CH3\phantom{............}}\\
|\phantom{.............................}|\phantom{...........}\\
\ce{CH3 - C - Br +\underset{\text{Nucleophile}}{OH-} ->CH3 - C - OH +\underset{\text{Bromide ion}}{Br-}}\\
|\phantom{.............................}|\phantom{...........}\\
\ce{\underset{\text{Tert-Butyl bromide}}{CH3}\phantom{.................}}
\ce{\underset{\text{tert−Butyl alcohol}}{CH3}}\phantom{.........}\\
\end{array}\]
Rate = k [(CH3)3C − Br]
ii. The reaction follows the first-order kinetics. That is, the rate of this reaction depends on the concentration of only one species, which is the substrate molecule, tert-butyl bromide. Hence, it is called substitution nucleophilic unimolecular, SN1 mechanism.
iii. It can be seen in the reaction that the concentration of the only substrate appears in the rate equation that is, the concentration of the nucleophile does not influence the reaction rate.
iv. In other words, tert-butyl bromide reacts with hydroxide by a two steps mechanism.
In the slow step C–X bond in the substrate undergoes heterolysis and in the subsequent fast step, the nucleophile uses its electron pair to form a new bond with the carbon undergoing change.
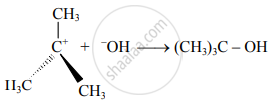
v. The SN1 mechanism is represented as,
- Step I:

- Step II:
APPEARS IN
संबंधित प्रश्न
from the following pair would undergo SN2 faster from the other?
Complete the following reaction giving major product.
\[\begin{array}{cc}\ce{CH3\phantom{................}}\\
|\phantom{...................}\\
\ce{CH3 - C - CH2 - Cl ->[Na/dry ether] A}\\
|\phantom{...................}\\
\ce{CH3\phantom{................}}
\end{array}\]
Arrange the following in the increasing order of boiling points.
- 1-Bromopropane
- 2- Bromopropane
- 1- Bromobutane
- 1-Bromo-2-methylpropane
Convert the following:
Benzyl alcohol to benzyl cyanide
Convert the following:
Ethanol to propane nitrile
Complete the following reaction sequence by writing the structural formulae of the organic compound 'A', 'B' and 'C'.
\[\ce{2-Bromobutane->[Alc.KOH]A->[][Br2]B->[][NaNH2]C}\]
Propane nitrile can be prepared by heating ____________
Major product of the following reaction is _______________
\[\ce{CH3-CH2-Mg-Br + NH3 ->?}\]
Nucleophilic substitution reaction of 2, 4-dinitrochlorobenzene is faster than p-nitrochlorobenzene. Give reason.
Write the correct order of increasing ease of dehydrohalogenation.
\[\ce{\underset{\text{(I)}}{CH3 - CH2 - CH2 - Cl}}\]
\[\ce{\underset{\text{(II)}}{CH3 - CH(Cl) - CH3}}\]
\[\begin{array}{cc}
\ce{CH3}\\
|\\
\ce{CH3-C-Cl}\phantom{...}\\
|\\
\ce{CH3}\\
\ce{(III)}
\end{array}\]
Explain. Aryl halides are less reactive than alkyl halides towards nucleophilic substitution reactions.
Explain the factors affecting SN1 and SN2 mechanism.
Which of the following is a primary halide?
Which of the following is least reactive towards SN1 reactions?
Dehydrohalogenation of an alkyl halide is ____________.
What type of hybridisation is present in carbocation formed during the alkaline hydrolysis of 1- bromo-1- phenyl ethane?
The SN2 reaction of a compound containing an asymmetric carbon atom always gives ____________.
Write the product formed when alkyl halide reacts with silver nitrite.
The major product formed when 2-bromobutane is treated with alcoholic KOH is ______.
Which of the following statements is incorrect regarding the dehydrohalogenation of alkenes?
Explain the dehydrohalogenation reaction of 2-chlorobutane.
Complete the following reaction giving major products.
\[\begin{array}{cc}
\ce{CH3}\phantom{.......}\\
|\phantom{.........}\\
\ce{CH3 - c - CH2 - Cl ->[Na/dry ether]}\\
|\phantom{.........}\\
\ce{CH3}\phantom{.......}
\end{array}\]
Complete the following reaction giving a major product.
\[\begin{array}{cc}
\ce{CH3}\phantom{.................}\\
|\phantom{...................}\\
\ce{CH3 - C - CH2 - Cl->[Na/dry ether]A}\\
|\phantom{..................}\\
\ce{CH3}\phantom{................}
\end{array}\]
Discuss β - elimination reaction.
Complete the following reactions giving major product.
\[\begin{array}{cc}
\ce{CH3}\phantom{................}\\
|\phantom{...................}\\
\ce{CH3 - C - CH2 - Cl ->[Na/dry ether] A}\\
|\phantom{...................}\\
\ce{CH3}\phantom{.................}
\end{array}\]
Complete the following reaction giving major product.
\[\begin{array}{cc}
\ce{CH3}\phantom{...............}\\
|\phantom{..................}\\
\ce{CH3 - C - CH2 - Cl->[Na/dry ether]A}\\
|\phantom{..................}\\
\ce{CH3}\phantom{...............}\\
\end{array}\]
