Advertisements
Advertisements
प्रश्न
Propane nitrile can be prepared by heating ____________
पर्याय
ethyl bromide with alcoholic KCN
propyl bromide with alcoholic KCN
ethyl bromide with alcoholic AgCN
propyl bromide with alcoholic AgCN
Advertisements
उत्तर
ethyl bromide with alcoholic KCN
Explanation:
\[\ce{\underset{\text{Ethyl bromide}}{CH3CH2Br}->[Alc.KCN,\Delta][-H2O]\underset{\text{Propane nitrile}}{CH3CH2CN}}\]
APPEARS IN
संबंधित प्रश्न
Name the reagent used to bring about the following conversion.
Ethyl bromide to ethyl isocyanide
Convert the following:
Ethanol to propane nitrile
Convert the following:
2-Chloropropane to propan-1-ol
The following will react faster by SN1 mechanism
Major product of the following reaction is _______________
\[\ce{CH3-CH2-Mg-Br + NH3 ->?}\]
Explain primary benzylic halide shows higher reactivity by SN1 mechanism than other primary alkyl halide.
What is dehydrohalogenation? State the rule for the formation of the preferred product of dehydrohalogenation.
Nitroalkanes are obtained in laboratory from primary or secondary alkyl halides by the action of ______.
\[\ce{C6H5CH2Cl + KCN(alc) -> X + Y}\]
Compounds X and Y are ____________.
Identify 'B' in the following reaction.
\[\ce{2-Bromobutane ->[KOH alc.][\Delta] A ->[HI] B}\]
Convert the following.
p-Nitrochlorobenzene to p-nitrophenol
Which of the following statements is incorrect regarding the dehydrohalogenation of alkenes?
With which halogen the reactions, of alkanes are explosive?
Complete the following reaction giving major products.
\[\begin{array}{cc}
\ce{CH3}\phantom{.......}\\
|\phantom{.........}\\
\ce{CH3 - c - CH2 - Cl ->[Na/dry ether]}\\
|\phantom{.........}\\
\ce{CH3}\phantom{.......}
\end{array}\]
Observe the following and answer the questions given below:
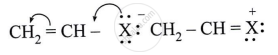
Name the type of halogen derivative.
Observe the following and answer the question given below:
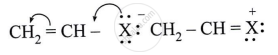
Comment on the bond length of C-X bond in it.
Discuss β - elimination reaction.
Complete the following reaction sequences by writing the structural formulae of the organic compounds 'A', 'B' and 'C'.
\[\ce{2-Bromobutan ->[alc. KOH] A ->[][Br2] B ->[][NANH2] C}\]
Complete the following reactions giving major product.
\[\begin{array}{cc}
\ce{CH3}\phantom{................}\\
|\phantom{...................}\\
\ce{CH3 - C - CH2 - Cl ->[Na/dry ether] A}\\
|\phantom{...................}\\
\ce{CH3}\phantom{.................}
\end{array}\]
Define and explain the SN1 mechanism with a suitable example.
What is the action of following on ethyl bromide?
alcoholic sodium hydroxide
Identify the chiral molecule from the following.
Complete the following reaction giving major product.
\[\begin{array}{cc}
\ce{CH3}\phantom{...............}\\
|\phantom{..................}\\
\ce{CH3 - C - CH2 - Cl ->[Na/dry ether]A}\\
|\phantom{...................}\\
\ce{CH3}\phantom{.................}
\end{array}\]
Complete the following reaction giving major product.
\[\begin{array}{cc}
\ce{CH3}\phantom{.................}\\
|\phantom{...................}\\
\ce{CH3 - C - CH2 - Cl ->[Na/dry ether] A}\\
|\phantom{...................}\\
\ce{CH3}\phantom{.................}
\end{array}\]
Complete the following reaction giving major product.
\[\begin{array}{cc}
\ce{CH3}\phantom{.................}\\
|\phantom{...................}\\
\ce{CH3 - C - CH2 - Cl ->[Na/dry ether] A}\\
|\phantom{...................}\\
\ce{CH3}\phantom{.................}\\
\end{array}\]
Which of the following reagent is used for conversion of alkyl halide into nitrile?
