Advertisements
Advertisements
प्रश्न
Convert the following:
2-Chloropropane to propan-1-ol
Advertisements
उत्तर
\[\begin{array}{cc}
\ce{H3C - CH - CH3 ->[Alc.KOH]\underset{Propene}{H3C - CH = CH2}->[HBr][Peroxide]\underset{1-Bromopropane}{H3C - CH2 - CH2Br}->[NaOH(aq.)]\underset{Propan-1-ol}{H3C - CH2 - CH2OH}}\\
|\phantom{...........................................................................................}\\
\ce{\underset{2-Chloropropane}{Cl}\phantom{..........................................................................................}}
\end{array}\]
APPEARS IN
संबंधित प्रश्न
Observe the following and answer the question given below.

Name the type of halogen derivative.
The following will react faster by SN1 mechanism
Major product of the following reaction is _______________
\[\ce{CH3-CH2-Mg-Br + NH3 ->?}\]
Explain the factors affecting SN1 and SN2 mechanism.
What is dehydrohalogenation? State the rule for the formation of the preferred product of dehydrohalogenation.
In alkaline hydrolysis of tertiary butyl bromide, ____________.
Ethyl bromide undergoes the following reaction:
\[\ce{\underset{Ethyl bromide}{C2H5Br} + \underset{(aq.)}{KOH} ->[\Delta] \underset{Ethyl alcohol}{C2H5OH} + KBr}\]
Which of the following is a WRONG statement?
Which one is most reactive towards nucleophilic addition reaction?
What type of hybridisation is present in carbocation formed during the alkaline hydrolysis of 1- bromo-1- phenyl ethane?
Which of the following is FALSE regarding SN2 reaction mechanism?
The SN2 reaction of a compound containing an asymmetric carbon atom always gives ____________.
Identify 'B' in the following reaction.
\[\ce{2-Bromobutane ->[KOH alc.][\Delta] A ->[HI] B}\]
The order of reactivities of the following alkyl halides for an SN2 reaction is ______.
Which of the following statements is incorrect regarding the dehydrohalogenation of alkenes?
Complete the following reaction giving major products.
\[\begin{array}{cc}
\ce{CH3}\phantom{.......}\\
|\phantom{.........}\\
\ce{CH3 - c - CH2 - Cl ->[Na/dry ether]}\\
|\phantom{.........}\\
\ce{CH3}\phantom{.......}
\end{array}\]
Observe the following and answer the question given below:
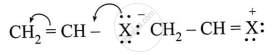
Can react by SN1 mechanism? Justify your answer.
Complete the following reaction sequences by writing the structural formulae of the organic compounds 'A', 'B' and 'C'.
\[\ce{2-Bromobutan ->[alc. KOH] A ->[][Br2] B ->[][NANH2] C}\]
Complete the following reactions giving major product.
\[\begin{array}{cc}
\ce{CH3}\phantom{................}\\
|\phantom{...................}\\
\ce{CH3 - C - CH2 - Cl ->[Na/dry ether] A}\\
|\phantom{...................}\\
\ce{CH3}\phantom{.................}
\end{array}\]
Complete the following reaction giving major product.
\[\begin{array}{cc}
\ce{CH3}\phantom{...............}\\
|\phantom{..................}\\
\ce{CH3 - C - CH2 - Cl->[Na/dry ether]A}\\
|\phantom{..................}\\
\ce{CH3}\phantom{...............}\\
\end{array}\]
Complete the reaction:
\[\ce{CH3CH2Cl ->[AgCN][alc.\Delta]}\] ?
Complete the following reaction giving major product.
\[\begin{array}{cc}
\ce{CH3}\phantom{................}\\
|\phantom{..................}\\
\ce{CH3 - C - CH2 - Cl ->[Na/dry ether]A}\\
|\phantom{..................}\\
\ce{CH3}\phantom{................}
\end{array}\]
Complete the following reaction giving major product.
\[\begin{array}{cc}
\ce{CH3}\phantom{................}\\
|\phantom{...................}\\
\ce{CH3 - C - CH2 - Cl->[Na/ dry ether]A}\\
|\phantom{...................}\\
\ce{CH3}\phantom{................}\\
\end{array}\]
Complete the following reaction giving major product.
\[\begin{array}{cc}
\ce{CH3}\phantom{.................}\\
|\phantom{...................}\\
\ce{CH3 - C - CH2 - Cl ->[Na/dry ether] A}\\
|\phantom{...................}\\
\ce{CH3}\phantom{.................}\\
\end{array}\]
Identify the substrate 'X' in the following reaction.
\[\ce{\underset{}{X + O2} ->[i) Co-naphtenate 423 K][ii) dil HClΔ] \underset{\underset{}{}}{Phenol + Acetone}}\]
