Advertisements
Advertisements
प्रश्न
Calculate the time required to heat 20 kg of water from 10°C to 35°C using an immersion heater rated 1000 W. Assume that 80% of the power input is used to heat the water. Specific heat capacity of water = 42000 J kg−1 K−1.
Advertisements
उत्तर
Given:-
Power rating of the immersion rod, P = 1000 W
Specific heat of water, S = 4200 J kg−1 K−1
Mass of water, M = 20 kg
Change in temperature, ΔT = 25 °C
Total heat required to raise the temperature of 20 kg of water from 10°C to 35°C is given by
Q = M × S × ΔT
Q = 20 × 4200 × 25
Q = 20 × 4200 × 25 = 21 × 105 J
Let the time taken to heat 20 kg of water from 10°C to 35°C be t. Only 80% of the immersion rod's heat is useful for heating water. Thus,
Energy of the immersion rod utilised for heating the water = t × (0.80) × 1000 J
t × (0.80) × 1000 J = 21 × 105 J
`t=(21xx10^5)/800=2625s`
`rArrt=2625/60=43.75minapprox44min`
APPEARS IN
संबंधित प्रश्न
Give reason of Water pipes burst in severe winter.
Explain the following:
How can you relate the formation of water droplets on the outer surface of a bottle taken out of refrigerator with formation of dew?
Explain the following:
In cold regions in winter, the rocks crack due to anomalous expansion of water.
A calorimeter contains 50 g of water at 50°C. The temperature falls to 45°C in 10 minutes. When the calorimeter contains 100 g of water at 50°C, it takes 18 minutes for the temperature to become 45°C. Find the water equivalent of the calorimeter.
Water is cooled from 4 °C to 0 °C. It will :
Explain, why are the exposed water pipes lagged with straw during severe winter?
A deep pond of water has its top layer frozen. What will be the likely temperature of water layer just in contact with ice?
The following figure shows a metal cylinder, containing boiling water. One half side A is polished and another half, B is painted black. Two thin metal sheets X and Y are painted black and have one rubber stopper fixed with wax on each sheet. These sheets are equidistant from the boiling water (container A, B) as shown in the diagram. What would you expect to happen after a few minutes? Give a reason for your answer.

What is the criterion of choosing the two metals for a bimetal strip?
Give reasons for the following:
Even when the water in the lakes is frozen, fish can survive.
_______ apparatus is used to study the anomalous behaviour of water.
Observe the following diagram and write the answers to the questions given below.
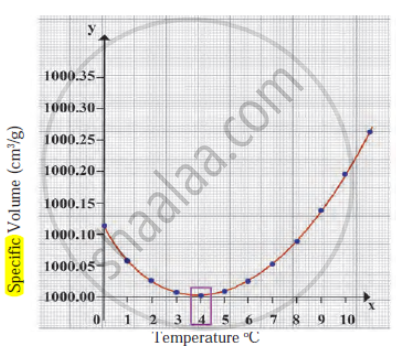
- Which process does the graph represent?
- What is the range of temperature responsible for the behaviour?
Name the following diagram appropriately.
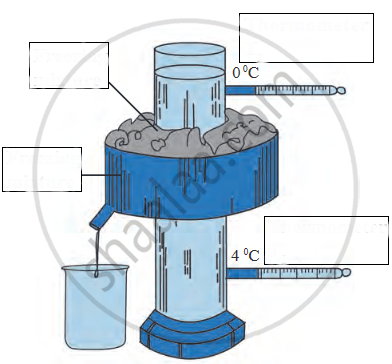
Write a short note.
Anomalous behaviour of water
