Advertisements
Advertisements
प्रश्न
Calculate the time required to heat 20 kg of water from 10°C to 35°C using an immersion heater rated 1000 W. Assume that 80% of the power input is used to heat the water. Specific heat capacity of water = 42000 J kg−1 K−1.
Advertisements
उत्तर
Given:-
Power rating of the immersion rod, P = 1000 W
Specific heat of water, S = 4200 J kg−1 K−1
Mass of water, M = 20 kg
Change in temperature, ΔT = 25 °C
Total heat required to raise the temperature of 20 kg of water from 10°C to 35°C is given by
Q = M × S × ΔT
Q = 20 × 4200 × 25
Q = 20 × 4200 × 25 = 21 × 105 J
Let the time taken to heat 20 kg of water from 10°C to 35°C be t. Only 80% of the immersion rod's heat is useful for heating water. Thus,
Energy of the immersion rod utilised for heating the water = t × (0.80) × 1000 J
t × (0.80) × 1000 J = 21 × 105 J
`t=(21xx10^5)/800=2625s`
`rArrt=2625/60=43.75minapprox44min`
APPEARS IN
संबंधित प्रश्न
On what basis and how will you determine whether air is saturated with vapour or not?
1 kg of ice at 0°C is mixed with 1 kg of steam at 100°C. What will be the composition of the system when thermal equilibrium is reached? Latent heat of fusion of ice = 3.36 × 103 J kg−1 and latent heat of vaporization of water = 2.26 × 106 J kg−1.
Explain the following
Fishes survive in ponds even when the atmospheric temperature is well below 0°C.
Density of water is maximum at :
Draw a graph between volume and temperature, when 5 cm3 of ice at -10°C is heated to form water at +10°C.
Explain, why are the exposed water pipes lagged with straw during severe winter?
Explain why do vegetables and fruits get damaged during severe frost?
Why does a thick glass tumbler crack when very hot water is poured in it?
What is the criterion of choosing the two metals for a bimetal strip?
Observe the given graph and answer the following questions:

- Name the process represented in the figure.
- At what temperature does this process take place?
_______ apparatus is used to study the anomalous behaviour of water.
While studying anomalous behaviour of water in Hope’s apparatus, the upper temperature of the thermometer : 0 °C : : lower temperature of the thermometer : _______
In cold regions, in winter the pipes for water supply break.
Draw a neat and labelled diagram of Hope’s apparatus.
Observe the following diagram and write the answers to the questions given below.
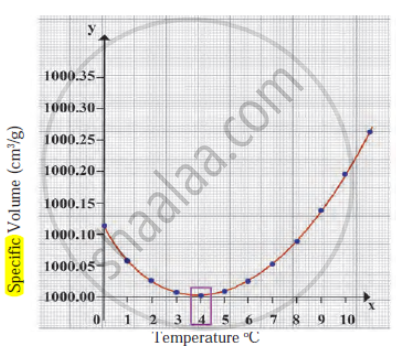
- Which process does the graph represent?
- What is the range of temperature responsible for the behaviour?
Match the columns:
| Column ‘A’ | Column ‘B’ |
| The density of water is maximum at | (a) 0°C |
| (b) 4°C | |
| (c) 100°C |
