Advertisements
Advertisements
प्रश्न
1 kg of ice at 0°C is mixed with 1 kg of steam at 100°C. What will be the composition of the system when thermal equilibrium is reached? Latent heat of fusion of ice = 3.36 × 103 J kg−1 and latent heat of vaporization of water = 2.26 × 106 J kg−1.
Advertisements
उत्तर
Given:-
Amount of ice at 0oC = 1 kg
Amount of steam at 100oC = 1 kg
Latent heat of fusion of ice = 3.36 × 103 J kg−1
Latent heat of vapourisation of water = 2.26 × 106 J kg−1
We can observe that the latent heat of fusion of ice (3.36 × 105 J kg−1) is smaller that latent heat of vapouisation of water (2.26 × 106 ). Therefore, ice will first change into water as less heat is required for this and there will be equilibrium between steam and water.
Heat absorbed by the ice when it changes into water (Q1) = 1×(3.36 × 105) J
Heat absorbed by the water formed to change its temperature from 0oC to 100oC (Q2) = 1 × 4200 × 100 = 4.2 × 105 J
Total heat absorbed by the ice to raise the temperature to 100°C, Q = Q1+Q2 = 3.36 × 105+ 4.2 × 105 = (3.36 + 4.2) × 105 = 7.56 × 105 J
The heat required to change ice into water at 100oC is supplied by the steam. This heat will be released by the steam and will then change into water.
If all the steam gets converted into water, heat released by steam, Q' = 1 ×( 2.26 × 106) J = 2.26 × 106 J
Amount of heat released is more than that required by the ice to get converted into water at 100oC. Thus,
Extra heat = Q − Q'
= (2.26 − 0.756) × 106
= 1.506 × 106
Let the mass of steam that is condensed into water be m. Thus,
`m=(7.56xx10^5)/(2.26xx10^6)=0.335kg=335gm`
Total amount of water at 100°C = 1000 + 335 = 1335 g =1.335 g
Steam left = 1− 0.335 = 0.665 kg = 665 gm
APPEARS IN
संबंधित प्रश्न
Water is used as a cooling agent.
The amount of water vapour in air is determined in terms of its ______.
Calculate the time required to heat 20 kg of water from 10°C to 35°C using an immersion heater rated 1000 W. Assume that 80% of the power input is used to heat the water. Specific heat capacity of water = 42000 J kg−1 K−1.
On a winter day the temperature of the tap water is 20°C whereas the room temperature is 5°C. Water is stored in a tank of capacity 0.5 m3 for household use. If it were possible to use the heat liberated by the water to lift a 10 kg mass vertically, how high can it be lifted as the water comes to the room temperature? Take g = 10 m s−2.
Draw a graph to show the variation in density of water with temperature in the range from 0°C to 10°C.
Describe an experiment to show that water has maximum density at 4°C. What important consequences follow this peculiar property of water? Discuss the importance of this phenomenon in nature.
Explain the following
A glass bottle completely filled with water and tightly closed at room temperature is likely to burst when kept in the freezer of a refrigerator.
Water is cooled from 4 °C to 0 °C. It will :
Density of water is maximum at :
Draw a graph between volume and temperature, when 5 cm3 of ice at -10°C is heated to form water at +10°C.
A deep pond of water has its top layer frozen. What will be the likely temperature of water layer at the bottom of the pond?
In cold regions, in winter the pipes for water supply break.
Draw a neat and labelled diagram of Hope’s apparatus.
Observe the following diagram and write the answers to the questions given below.
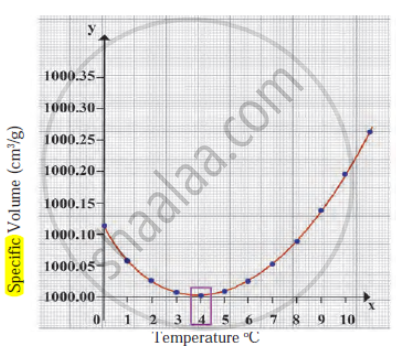
- Which process does the graph represent?
- What is the range of temperature responsible for the behaviour?
Match the columns:
| Column ‘A’ | Column ‘B’ |
| The density of water is maximum at | (a) 0°C |
| (b) 4°C | |
| (c) 100°C |
A graph between the volume and temperature of water is shown. Explain the anomalous behaviour of water.

