Advertisements
Advertisements
Question
1 kg of ice at 0°C is mixed with 1 kg of steam at 100°C. What will be the composition of the system when thermal equilibrium is reached? Latent heat of fusion of ice = 3.36 × 103 J kg−1 and latent heat of vaporization of water = 2.26 × 106 J kg−1.
Advertisements
Solution
Given:-
Amount of ice at 0oC = 1 kg
Amount of steam at 100oC = 1 kg
Latent heat of fusion of ice = 3.36 × 103 J kg−1
Latent heat of vapourisation of water = 2.26 × 106 J kg−1
We can observe that the latent heat of fusion of ice (3.36 × 105 J kg−1) is smaller that latent heat of vapouisation of water (2.26 × 106 ). Therefore, ice will first change into water as less heat is required for this and there will be equilibrium between steam and water.
Heat absorbed by the ice when it changes into water (Q1) = 1×(3.36 × 105) J
Heat absorbed by the water formed to change its temperature from 0oC to 100oC (Q2) = 1 × 4200 × 100 = 4.2 × 105 J
Total heat absorbed by the ice to raise the temperature to 100°C, Q = Q1+Q2 = 3.36 × 105+ 4.2 × 105 = (3.36 + 4.2) × 105 = 7.56 × 105 J
The heat required to change ice into water at 100oC is supplied by the steam. This heat will be released by the steam and will then change into water.
If all the steam gets converted into water, heat released by steam, Q' = 1 ×( 2.26 × 106) J = 2.26 × 106 J
Amount of heat released is more than that required by the ice to get converted into water at 100oC. Thus,
Extra heat = Q − Q'
= (2.26 − 0.756) × 106
= 1.506 × 106
Let the mass of steam that is condensed into water be m. Thus,
`m=(7.56xx10^5)/(2.26xx10^6)=0.335kg=335gm`
Total amount of water at 100°C = 1000 + 335 = 1335 g =1.335 g
Steam left = 1− 0.335 = 0.665 kg = 665 gm
APPEARS IN
RELATED QUESTIONS
The amount of water vapour in air is determined in terms of its ______.
Explain the following:
How can you relate the formation of water droplets on the outer surface of a bottle taken out of refrigerator with formation of dew?
Draw a graph to show the variation in density of water with temperature in the range from 0°C to 10°C.
Explain, why are the exposed water pipes lagged with straw during severe winter?
A deep pond of water has its top layer frozen. What will be the likely temperature of water layer just in contact with ice?
A deep pond of water has its top layer frozen. What will be the likely temperature of water layer at the bottom of the pond?
In cold countries, the water pipes are covered with poor conductors. Why?
The following figure shows a metal cylinder, containing boiling water. One half side A is polished and another half, B is painted black. Two thin metal sheets X and Y are painted black and have one rubber stopper fixed with wax on each sheet. These sheets are equidistant from the boiling water (container A, B) as shown in the diagram. What would you expect to happen after a few minutes? Give a reason for your answer.

Give reasons for the following:
Even when the water in the lakes is frozen, fish can survive.
Observe the given graph and answer the following questions:

- Name the process represented in the figure.
- At what temperature does this process take place?
In a region with a cold climate the aquatic animals can survive at 4 °C, because _______.
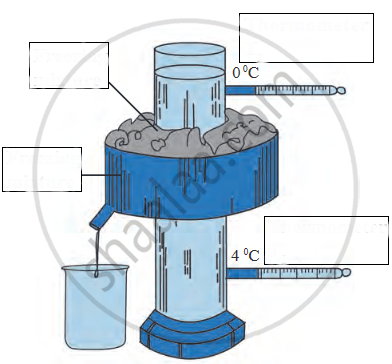
_______ apparatus is used to study the anomalous behaviour of water.
While studying anomalous behaviour of water in Hope’s apparatus, the upper temperature of the thermometer : 0 °C : : lower temperature of the thermometer : _______
Write the name.
The instrument used to study anomalous behaviour of water.
Observe the following diagram and write the answers to the questions given below.
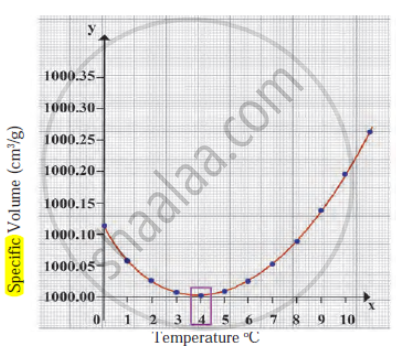
- Which process does the graph represent?
- What is the range of temperature responsible for the behaviour?
Name the following diagram appropriately.
Observe the given picture and answer the following questions.
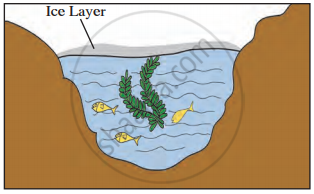
- Which property do you understand in this picture?
- What is the temperature of the water at the surface?
- What is the temperature below the layer of ice on the surface?
