Advertisements
Advertisements
प्रश्न
On a winter day the temperature of the tap water is 20°C whereas the room temperature is 5°C. Water is stored in a tank of capacity 0.5 m3 for household use. If it were possible to use the heat liberated by the water to lift a 10 kg mass vertically, how high can it be lifted as the water comes to the room temperature? Take g = 10 m s−2.
Advertisements
उत्तर
Given:-
Initial temperature of the water, Ti = 20°C
Final temperature of the water (room temperature), Tf = 5°C
Change in temeprature, ΔT = 20°C − 5°C = 15°C
Volume of water = 0.5 m3
Density of water, d = 1000 kg/m3
Mass of the water, M = (0.5 × 1000) kg = 500 kg
Heat liberated as the temperature of water changes from 20°C to 5°C is given by
Q = M×S×ΔT
Q = (500 × 4200 × 15) J
Q = (500 × 4200 × 15) J
Q = (75 × 420 × 1000) J
Q = 31500 × 1000 J = 315×105 J
Let the height to which the mass is lifted be h.
The energy required to lift the block = mgh = 10 × 10 × h = 100 h
Acording to the question,
Q = mgh
⇒ 100 h = 315×105 J
⇒ h = 315×103 m = 315 km
APPEARS IN
संबंधित प्रश्न
Give reason for Sea water does not freeze at 0°C.
The amount of water vapour in air is determined in terms of its ______.
What is the role of the anomalous behaviour of water in preserving aquatic life in regions of cold climate?
1 kg of ice at 0°C is mixed with 1 kg of steam at 100°C. What will be the composition of the system when thermal equilibrium is reached? Latent heat of fusion of ice = 3.36 × 103 J kg−1 and latent heat of vaporization of water = 2.26 × 106 J kg−1.
Calculate the time required to heat 20 kg of water from 10°C to 35°C using an immersion heater rated 1000 W. Assume that 80% of the power input is used to heat the water. Specific heat capacity of water = 42000 J kg−1 K−1.
Draw a graph to show the variation in density of water with temperature in the range from 0°C to 10°C.
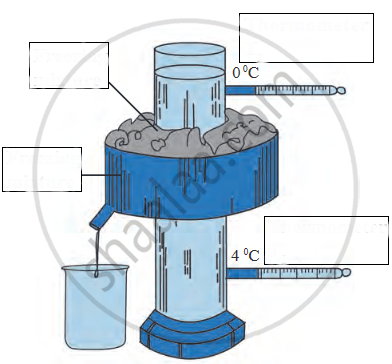
Describe an experiment to show that water has maximum density at 4°C. What important consequences follow this peculiar property of water? Discuss the importance of this phenomenon in nature.
Explain the following
A glass bottle completely filled with water and tightly closed at room temperature is likely to burst when kept in the freezer of a refrigerator.
The following figure shows a metal cylinder, containing boiling water. One half side A is polished and another half, B is painted black. Two thin metal sheets X and Y are painted black and have one rubber stopper fixed with wax on each sheet. These sheets are equidistant from the boiling water (container A, B) as shown in the diagram. What would you expect to happen after a few minutes? Give a reason for your answer.

Study the following diagrams and write down your observations.

Observe the given graph and answer the following questions:

- Name the process represented in the figure.
- At what temperature does this process take place?
When the temperature of water decreases below 4 °C it’s volume _______.
_______ apparatus is used to study the anomalous behaviour of water.
Write the name.
The instrument used to study anomalous behaviour of water.
Write scientific reason.
Placing a plastic bottle filled with water in the freezing compartment in the freezer can cause the bottle to explode.
Observe the following diagram and write the answers to the questions given below.
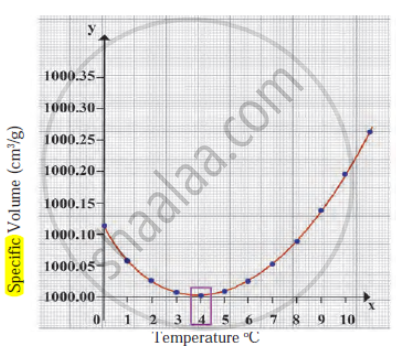
- Which process does the graph represent?
- What is the range of temperature responsible for the behaviour?
Name the following diagram appropriately.