Advertisements
Advertisements
प्रश्न
A calorimeter contains 50 g of water at 50°C. The temperature falls to 45°C in 10 minutes. When the calorimeter contains 100 g of water at 50°C, it takes 18 minutes for the temperature to become 45°C. Find the water equivalent of the calorimeter.
Advertisements
उत्तर
Let water equivalent to calorimeter be w.
Change in temperature = 5°C
Specific heat of water = 4200 J/Kg °C
Rate of flow of heat is given by
q = Energy per unit time = `(msDeltaT)/t`
Case 1:
`q_1 = ((w +50 xx 10^-3 )xx 4200xx5)/10`
Case 2
`q^2 = ((w+100 xx10^-3)xx 4200xx5)/18`
From calorimeter theory, these two rates of flow of heat should be equal to each other.
⇒ q1 = q2
`((w + 50 xx 10^-3)xx 4200xx5)/10 = ((w + 100xx10^-3)xx4200xx5)/18`
`rArr 18(W + 50 xx 10^-3) = 10(W + 100 xx10^-3)`
⇒ W = 12.5 ×10-3 kg
⇒ W = 12.5 g
APPEARS IN
संबंधित प्रश्न
What is the role of the anomalous behaviour of water in preserving aquatic life in regions of cold climate?
On a winter day the temperature of the tap water is 20°C whereas the room temperature is 5°C. Water is stored in a tank of capacity 0.5 m3 for household use. If it were possible to use the heat liberated by the water to lift a 10 kg mass vertically, how high can it be lifted as the water comes to the room temperature? Take g = 10 m s−2.
What do you mean by the anomalous expansion of water?
Explain the following
Fishes survive in ponds even when the atmospheric temperature is well below 0°C.
Explain the following
A hollow glass sphere which floats with its entire volume submerged in water at 4°C, sinks when water is heated above 4°C.
Explain the following
A glass bottle completely filled with water and tightly closed at room temperature is likely to burst when kept in the freezer of a refrigerator.
Draw a graph between volume and temperature, when 5 cm3 of ice at -10°C is heated to form water at +10°C.
A deep pond of water has its top layer frozen. What will be the likely temperature of water layer at the bottom of the pond?
Why does a thick glass tumbler crack when very hot water is poured in it?
Give reasons for the following:
Even when the water in the lakes is frozen, fish can survive.
Observe the given graph and answer the following questions:

- Name the process represented in the figure.
- At what temperature does this process take place?
_______ apparatus is used to study the anomalous behaviour of water.
Observe the following diagram and write the answers to the questions given below.
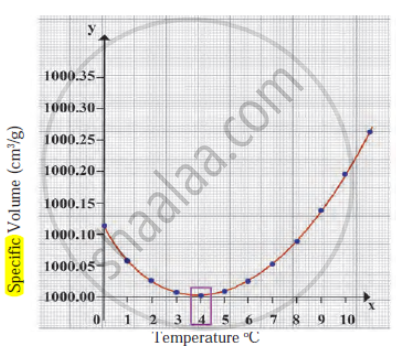
- Which process does the graph represent?
- What is the range of temperature responsible for the behaviour?
Write a short note.
Anomalous behaviour of water
