Advertisements
Advertisements
प्रश्न
A calorimeter contains 50 g of water at 50°C. The temperature falls to 45°C in 10 minutes. When the calorimeter contains 100 g of water at 50°C, it takes 18 minutes for the temperature to become 45°C. Find the water equivalent of the calorimeter.
Advertisements
उत्तर
Let water equivalent to calorimeter be w.
Change in temperature = 5°C
Specific heat of water = 4200 J/Kg °C
Rate of flow of heat is given by
q = Energy per unit time = `(msDeltaT)/t`
Case 1:
`q_1 = ((w +50 xx 10^-3 )xx 4200xx5)/10`
Case 2
`q^2 = ((w+100 xx10^-3)xx 4200xx5)/18`
From calorimeter theory, these two rates of flow of heat should be equal to each other.
⇒ q1 = q2
`((w + 50 xx 10^-3)xx 4200xx5)/10 = ((w + 100xx10^-3)xx4200xx5)/18`
`rArr 18(W + 50 xx 10^-3) = 10(W + 100 xx10^-3)`
⇒ W = 12.5 ×10-3 kg
⇒ W = 12.5 g
APPEARS IN
संबंधित प्रश्न
Give reason of Water pipes burst in severe winter.
How does the anomalous expansion of water help aquatic organisms in cold climates?
The amount of water vapour in air is determined in terms of its ______.
What is the role of the anomalous behaviour of water in preserving aquatic life in regions of cold climate?
Explain the following:
How can you relate the formation of water droplets on the outer surface of a bottle taken out of refrigerator with formation of dew?
Consider the situation of the previous problem. Assume that the temperature of the water at the bottom of the lake remains constant at 4°C as the ice forms on the surface (the heat required to maintain the temperature of the bottom layer may come from the bed of the lake). The depth of the lake is 1.0 m. Show that the thickness of the ice formed attains a steady state maximum value. Find this value. The thermal conductivity of water = 0.50 W m−1°C−1. Take other relevant data from the previous problem.
At what temperature is the density of water is maximum? State its value.
Explain the following
A hollow glass sphere which floats with its entire volume submerged in water at 4°C, sinks when water is heated above 4°C.
Explain, why does a glass bottle completely filled with water and tightly capped burst when placed in a freezer?
Explain why do vegetables and fruits get damaged during severe frost?
A deep pond of water has its top layer frozen. What will be the likely temperature of water layer at the bottom of the pond?
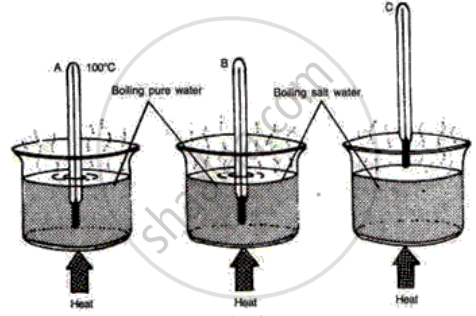
The following diagrams illustrate three situations involving thermometers which are labeled A, Band C. In each situation the thermometers indicate different readings.
(i) What do you expect the approximate reading of the thermometer B and C would be? Give a reason for your answer.
(ii) How would the readings of A and B help you in calibrating a thermometer?
What is the criterion of choosing the two metals for a bimetal strip?
When the temperature of water decreases below 4 °C it’s volume _______.
Write scientific reason.
Fish can survive even in frozen ponds in cold regions.
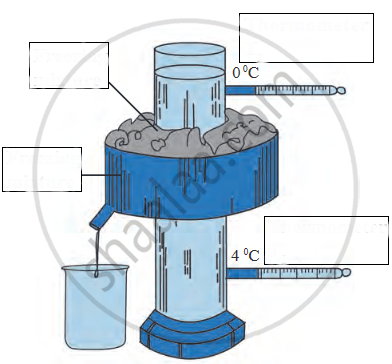
Name the following diagram appropriately.