Advertisements
Advertisements
प्रश्न
Study the following diagrams and write down your observations.

Advertisements
उत्तर

In the diagram, it is clear that the ice will melt faster in the flask than in the test tube because water is a bad conductor of heat and the transfer of heat in liquid through convection is from bottom to top.
APPEARS IN
संबंधित प्रश्न
Give reason of Water pipes burst in severe winter.
Consider the situation of the previous problem. Assume that the temperature of the water at the bottom of the lake remains constant at 4°C as the ice forms on the surface (the heat required to maintain the temperature of the bottom layer may come from the bed of the lake). The depth of the lake is 1.0 m. Show that the thickness of the ice formed attains a steady state maximum value. Find this value. The thermal conductivity of water = 0.50 W m−1°C−1. Take other relevant data from the previous problem.
Explain, why are the exposed water pipes lagged with straw during severe winter?
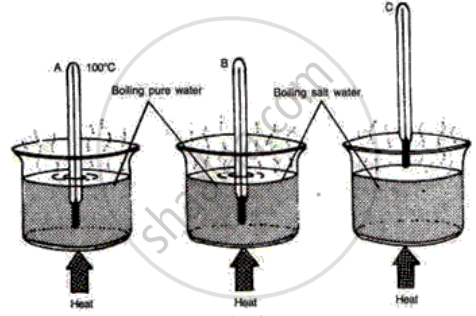
The following diagrams illustrate three situations involving thermometers which are labeled A, Band C. In each situation the thermometers indicate different readings.
(i) What do you expect the approximate reading of the thermometer B and C would be? Give a reason for your answer.
(ii) How would the readings of A and B help you in calibrating a thermometer?
The following figure shows a metal cylinder, containing boiling water. One half side A is polished and another half, B is painted black. Two thin metal sheets X and Y are painted black and have one rubber stopper fixed with wax on each sheet. These sheets are equidistant from the boiling water (container A, B) as shown in the diagram. What would you expect to happen after a few minutes? Give a reason for your answer.

Observe the given graph and answer the following questions:

- Name the process represented in the figure.
- At what temperature does this process take place?
