Advertisements
Advertisements
The following graph shows the variation of photocurrent for a photosensitive metal :
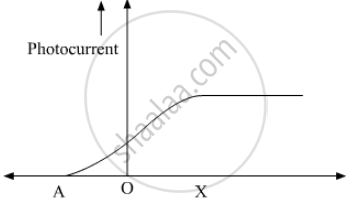
(a) Identify the variable X on the horizontal axis.
(b) What does the point A on the horizontal axis represent?
(c) Draw this graph for three different values of frequencies of incident radiation v1, v2 and v3 (v1 > v2 > v3) for same intensity.
(d) Draw this graph for three different values of intensities of incident radiation I1, I2 and I3 (I1 > I2 > I3) having same frequency.
Concept: Experimental Study of Photoelectric Effect
The work function of the following metals is given : Na 2.75 ev, K = 2.3 eV, Mo = 4.17 eV and Ni = 5.15 eV. Which of these metals will not cause photoelectric emission for radiation of wavelength 3300 Å from a laser source placed 1 m away from these metals? What happens if the laser source is brought nearer and placed 50 cm away?
Concept: Electron Emission
A electron of mass me revolves around a nucleus of charge +Ze. Show that it behaves like a tiny magnetic dipole. Hence prove that the magnetic moment associated wit it is expressed as `vecμ =−e/(2 m_e)vecL `, where `vec L` is the orbital angular momentum of the electron. Give the significance of negative sign.
Concept: Wave Nature of Matter
An α-particle and a proton are accelerated through the same potential difference. Find the ratio of their de Broglie wavelength.
Concept: de-Broglie Relation
Radiation of frequency 1015 Hz is incident on two photosensitive surface P and Q. There is no photoemission from surface P. Photoemission occurs from surface Q but photoelectrons have zero kinetic energy. Explain these observations and find the value of work function for surface Q.
Concept: Einstein’s Equation - Particle Nature of Light
An electron, an alpha particle and a proton have the same kinetic energy.
Which one of these particles has the largest de-Broglie wavelength?
Concept: de-Broglie Relation
The ground state energy of hydrogen atom is – 13∙6 eV. If an electron makes a transition from an energy level – 1∙51 eV to – 3∙4 eV, calculate the wavelength of the spectral line emitted and name the series of hydrogen spectrum to which it belongs.
Concept: Hydrogen Spectrum
Find out the wavelength of the electron orbiting in the ground state of hydrogen atom.
Concept: Hydrogen Spectrum
Find the wavelength of the electron orbiting in the first excited state in hydrogen atom.
Concept: The Line Spectra of the Hydrogen Atom
The energy levels of an atom are as shown below. Which of them will result in the transition of a photon of wavelength 275 nm?
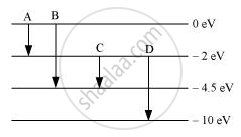
Concept: Energy Levels
Which transition corresponds to emission of radiation of maximum wavelength?
Concept: Energy Levels
Two nuclei have mass numbers in the ratio 1: 2. What is the ratio of their nuclear densities?
Concept: Atomic Masses and Composition of Nucleus
A radioactive nucleus ‘A’ undergoes a series of decays according to the following scheme:
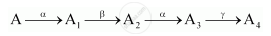
The mass number and atomic number of A are 180 and 72 respectively. What are these numbers for A4?
Concept: Law of Radioactive Decay
The radioactive isotope D decays according to the sequence
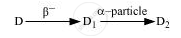
If the mass number and atomic number of D2 are 176 and 71 respectively, what is (i) the mass number (ii) atomic number of D?
Concept: Law of Radioactive Decay
Derive an expression for voltage gain of the amplifier and hence show that the output voltage is in opposite phase with the input voltage.
Concept: Junction Transistor >> Transistor as an Amplifier (Ce-configuration)
In the following diagram 'S' is a semiconductor. Would you increase or decrease the value of R to keep the reading of the ammeter A constant when S is heated? Give reason for your answer.

Concept: Semiconductor Diode
State its any ‘two’ uses of photodiode.
Concept: Special Purpose P-n Junction Diodes
Draw its I – V characteristics of photodiode
Concept: Semiconductor Diode
A zener diode is fabricated by heavily doping both p- and n- sides of the junction. Explain, why?
Concept: p-n Junction
Explain briefly with the help of necessary diagrams, the forward biasing of a p-n junction diode. Also draw characteristic curves.
Concept: p-n Junction
