Advertisements
Advertisements
प्रश्न
Give scientific reasons for the following:
Atomic radius goes on decreasing while going from left to right in a period.
Give scientific reasons:
While going from left to right within a period, the size of the atom decreases.
Advertisements
उत्तर
- While going from left to right within a period, the atomic number increases one by one, meaning the positive charge on the nucleus increases by one unit at a time.
- However, the additional electron gets added to the same outermost shell. Due to the increased nuclear charge, the electrons are pulled towards the nucleus to a greater extent, and thereby the size of the atom decreases.
APPEARS IN
संबंधित प्रश्न
Answer the following in respect of element `31/15 P`
Give its electronic configuration
Answer the following in respect of element `31/15 P `
Give its formula with chlorine?
What do you understand by atomic size? State its unit?
Give the trends in atomic size on moving down the group.
Give the trends in atomic size on moving across the period left to right.
Fill in the blank:
On moving across a period from right to left in the periodic table, the atomic size of the atom ___________.
Write the name and symbol of the element from the description.
The noble gas with the smallest atomic radius.
Arrange the following in increasing order of property indicated
I, I+, I- (atomic size)
With reference to the variation of properties in the Periodic Table, which of the following is generally true?
Atomic size increases from left to right across a period.
Supply the missing word from those in the brackets:
If an element has seven electrons in its outermost shell then it is likely to have the ______ (largest/ smallest) atomic size among all the elements in the same period.
While going from top to bottom in a group the atomic radius _______.
Write an Explanation.
Atomic radius
Which of the following gives the correct increasing order of the atomic radii of O, F, and N?
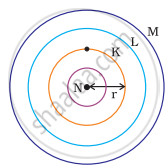
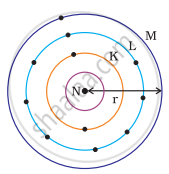
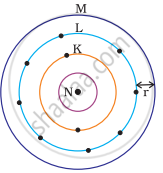
Which one of the following depict the correct representation of atomic radius(r) of an atom?
|
(i) |
(ii) |
|
(iii) |
(iv) |
Elements have been arranged in the following sequence on the basis of their increasing atomic masses.
| F, | Na, | Mg, | Al, | Si, | P, | S, | Cl, | Ar, | K |
- Pick two sets of elements which have similar properties.
- The given sequence represents which law of classification of elements?
- In below ladder symbols of elements are jumbled up. Rearrange these symbols of elements in the increasing order of their atomic number in the Periodic Table.
- Arrange them in the order of their group also.
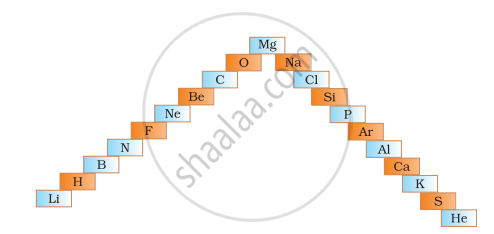
An element with the largest atomic radius among the following is ______.
Give reason for the following:
The size of a Cl− ion is greater than the size of a Cl atom.