Advertisements
Advertisements
प्रश्न
What happens to the atomic size of elements on moving from left to right in a period?
Advertisements
उत्तर
Atomic size of elements decreases on moving from left to right in a period.
APPEARS IN
संबंधित प्रश्न
Which is greater in size Fe2+ or Fe3+?
Give reason for the following:
Argon atom is bigger than chlorine atom.
Supply the missing word from those in the brackets:
If an element has seven electrons in its outermost shell then it is likely to have the ______ (largest/ smallest) atomic size among all the elements in the same period.
_______ is the distance between the nucleus of the atom and its outermost shell.
Some elements and their atomic radii are given here. Arrange them in decreasing order of their atomic radii. Identify which of the above elements is the biggest atom and which is smallest?
| Element | K | Na | Rb | Cs | Li |
| Atomic radius (pm) | 231 | 186 | 244 | 262 | 151 |
Which among the following elements has the largest atomic radii?
Write the formula of the product formed when the element A (atomic number 19) combines with the element B (atomic number 17). Draw its electronic dot structure. What is the nature of the bond formed?
- Electropositive nature of the element(s) increases down the group and decreases across the period
- Electronegativity of the element decreases down the group and increases across the period
- Atomic size increases down the group and decreases across a period (left to right)
- Metallic character increases down the group and decreases across a period.
On the basis of the above trends of the Periodic Table, answer the following about the elements with atomic numbers 3 to 9.
- Name the most electropositive element among them
- Name the most electronegative element
- Name the element with smallest atomic size
- Name the element which is a metalloid
- Name the element which shows maximum valency.
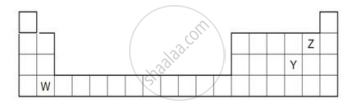
The diagram below shows part of the periodic table.
- Which elements would react together to form covalent compounds?
- Between the two elements W and Z, which will have a bigger atomic radius? Why?
Which one of the following has the largest atomic radius?
