Advertisements
Advertisements
प्रश्न
Arrange the following as per the instruction given in the bracket
Na, K, Li (Increasing atomic size)
Advertisements
उत्तर
Li < Na < K
APPEARS IN
संबंधित प्रश्न
Answer the following in respect of element `31/15 P`
Give its electronic configuration
Answer the following in respect of element `31/15 P `
Is it a reducing agent or oxidizing agent?
Which is greater in size Fe2+ or Fe3+?
Fill in the blank:
On moving across a period from right to left in the periodic table, the atomic size of the atom ___________.
Give reasons for the following:
The size of the anion is greater than the size of the parent atom.
What happens to the atomic size of elements on moving from left to right in a period?
The changes in the properties of elements on moving from left to right across a period of the Periodic Table. For the property, choose the correct answer.
The atomic size:
Which of the following is the correct order of size?
Which of the following gives the correct increasing order of the atomic radii of O, F, and N?
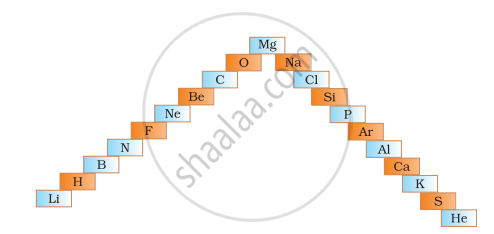
- In below ladder symbols of elements are jumbled up. Rearrange these symbols of elements in the increasing order of their atomic number in the Periodic Table.
- Arrange them in the order of their group also.