Advertisements
Advertisements
प्रश्न
Arrange the following as per the instruction given in the bracket:
Carbon, Fluorine, Beryllium (decreasing order of atomic size).
Advertisements
उत्तर
Beryllium > Carbon > Fluorine
Explanation:
As we proceed along a period, the size of the atom decreases due to increased nuclear pull.
APPEARS IN
संबंधित प्रश्न
What do you understand by atomic size? State its unit?
Give the trends in atomic size on moving across the period left to right.
Which is greater in size Fe2+ or Fe3+?
Size of atom progressively becomes smaller when we move from sodium (Na) to chlorine (CI) in the third period of the periodic table?
Write the name and symbol of the element from the description.
The atom having the smallest size.
What happens to the atomic size of elements on moving from left to right in a period?
Which of the following is the correct order of atomic size?
Which of the following are the characteristics of isotopes of an element?
- Isotopes of an element have same atomic masses
- Isotopes of an element have same atomic number
- Isotopes of an element show same physical properties
- Isotopes of an element show same chemical properties
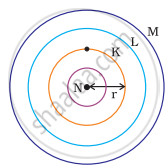
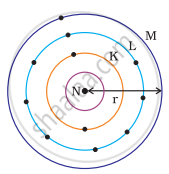
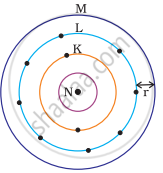
Which one of the following depict the correct representation of atomic radius(r) of an atom?
|
(i) |
(ii) |
|
(iii) |
(iv) |
This question refers to the elements of the Periodic Table with atomic numbers from 3 to 18. Some of the elements are shown by letters, but the letters are not the usual symbols of the elements.
| 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 |
| A | B | C | D | E | F | G | H |
| 11 | 12 | 13 | 14 | 15 | 16 | 17 | 18 |
| I | J | K | L | M | N | O | P |
Which of these have least atomic size in period 3?