Advertisements
Advertisements
प्रश्न
Which is greater in size an atom or a cation?
Advertisements
उत्तर
An atom is always greater than a cation since the loss of electrons forms a cation; hence, protons are more than electrons in a cation. The nucleus strongly attracts electrons and pulls them inward.
APPEARS IN
संबंधित प्रश्न
Give reason for the following:
Argon atom is bigger than chlorine atom.
Supply the missing word from those in the brackets:
If an element has seven electrons in its outermost shell then it is likely to have the ______ (largest/ smallest) atomic size among all the elements in the same period.
While going from top to bottom in a group the atomic radius _______.
Nanometer unit is used to measure atomic radius.
Write information about the given atomic numbers in the table. 10, 20, 7.
| Atomic Number | Electronic configuration | Group | Period | Element |
| 10 | ||||
| 20 | ||||
| 7 |
Which of the following is the correct order of atomic size?
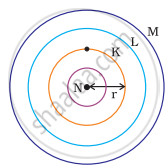
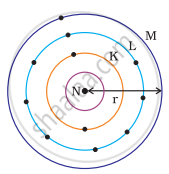
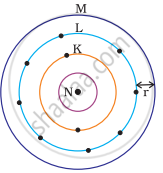
Which one of the following depict the correct representation of atomic radius(r) of an atom?
|
(i) |
(ii) |
|
(iii) |
(iv) |
Atomic number of a few elements are given below
10, 20, 7, 14
- Identify the elements
- Identify the Group number of these elements in the Periodic Table
- Identify the Periods of these elements in the Periodic Table
- What would be the electronic configuration for each of these elements?
- Determine the valency of these elements
Arrange the following in order of increasing radii:
Cl−, Cl
Explain your choice.
This question refers to the elements of the Periodic Table with atomic numbers from 3 to 18. Some of the elements are shown by letters, but the letters are not the usual symbols of the elements.
| 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 |
| A | B | C | D | E | F | G | H |
| 11 | 12 | 13 | 14 | 15 | 16 | 17 | 18 |
| I | J | K | L | M | N | O | P |
Which of these have least atomic size in period 3?