Advertisements
Advertisements
प्रश्न
Nanometer unit is used to measure atomic radius.
विकल्प
Right
Wrong
Advertisements
उत्तर
Nanometer unit is used to measure atomic radius- Wrong
APPEARS IN
संबंधित प्रश्न
What do you understand by atomic size? State its unit?
Give the trends in atomic size on moving down the group.
Give reason for the following:
Argon atom is bigger than chlorine atom.
Write the name and symbol of the element from the description.
The atom having the smallest atomic mass.
What happens to the atomic size of elements on moving from left to right in a period?
With reference to the variation of properties in the Periodic Table, which of the following is generally true?
Atomic size increases from left to right across a period.
The metals of Group 2 from top to bottom are Be, Mg, Ca, Sr and Ba. Which one of these elements will form ions most readily and why?
Fill in the blank by selecting the correct word from the bracket.
If an element has seven electrons in its outermost shell then it is likely to have the _____ atomic size among all the elements in the same period.
The size of an atom is indicated by its _______.
_______ is the distance between the nucleus of the atom and its outermost shell.
Write an Explanation.
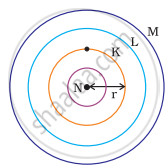
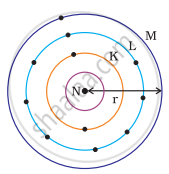
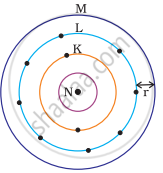
Atomic radius
Carbon belongs to the second period and Group 14. Silicon belongs to the third period and Group 14. If the atomic number of carbon is 6, the atomic number of silicon is ______
Which of the following is the correct order of atomic size?
Which of the following is the correct order of size?
Which of the following gives the correct increasing order of the atomic radii of O, F, and N?
When an atom of iodine becomes an iodine ion (I–) the radius will ______
Which one of the following depict the correct representation of atomic radius(r) of an atom?
|
(i) |
(ii) |
|
(iii) |
(iv) |
Write the formula of the product formed when the element A (atomic number 19) combines with the element B (atomic number 17). Draw its electronic dot structure. What is the nature of the bond formed?
An element X (atomic number 17) reacts with an element Y (atomic number 20) to form a divalent halide.
- Where in the periodic table are elements X and Y placed?
- Classify X and Y as metal (s), non-metal (s) or metalloid (s)
- What will be the nature of oxide of element Y? Identify the nature of bonding in the compound formed
- Draw the electron dot structure of the divalent halide
Arrange the following in order of increasing radii:
N, O, P
Explain your choice.