Advertisements
Advertisements
प्रश्न
Write the name and symbol of the element from the description.
The atom having the smallest atomic mass.
Advertisements
उत्तर
Name: Hydrogen
Symbol: H
संबंधित प्रश्न
Answer the following in respect of element `31/15 P `
Give its formula with chlorine?
Arrange the elements of second and third periods in increasing order of their atomic size (excluding noble gases).
Why is the size of sodium is greater than magnesium?
Which is greater in size Fe2+ or Fe3+?
Size of atom progressively becomes smaller when we move from sodium (Na) to chlorine (CI) in the third period of the periodic table?
Give reason for the following:
Argon atom is bigger than chlorine atom.
Write the name and symbol of the element from the description.
The atom having the smallest size.
The size of an atom is indicated by its _______.
Atomic radius is expressed in the unit _______.
While going from top to bottom in a group the atomic radius _______.
Moving from left to right, the size of the atom decreases.
Write scientific reason.
In same period, boron and oxygen elements have different atomic size.
Some elements and their atomic radii are given here. Arrange them in decreasing order of their atomic radii. Identify which of the above elements is the biggest atom and which is smallest?
| Element | K | Na | Rb | Cs | Li |
| Atomic radius (pm) | 231 | 186 | 244 | 262 | 151 |
When an atom of iodine becomes an iodine ion (I–) the radius will ______
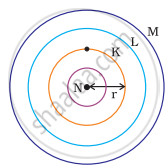
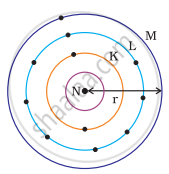
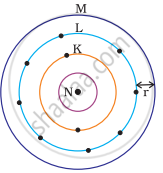
Which one of the following depict the correct representation of atomic radius(r) of an atom?
|
(i) |
(ii) |
|
(iii) |
(iv) |
An element X (atomic number 17) reacts with an element Y (atomic number 20) to form a divalent halide.
- Where in the periodic table are elements X and Y placed?
- Classify X and Y as metal (s), non-metal (s) or metalloid (s)
- What will be the nature of oxide of element Y? Identify the nature of bonding in the compound formed
- Draw the electron dot structure of the divalent halide
An element with the largest atomic radius among the following is ______.
Arrange the following in order of increasing radii:
Cl−, Cl
Explain your choice.
Which one has the largest size?