Advertisements
Advertisements
प्रश्न
Write an Explanation.
Atomic radius
Advertisements
उत्तर
Atomic radius is a measure of an atom’s size and changes predictably across periods and groups in the periodic table.
APPEARS IN
संबंधित प्रश्न
Arrange the following as per the instruction given in the brackets:
Na, K, Li (Increasing atomic size)
What do you understand by atomic size? State its unit?
Which is greater in size Fe2+ or Fe3+?
Fill in the blank:
On moving across a period from right to left in the periodic table, the atomic size of the atom ___________.
Arrange the following in order of increasing radii:
CI- , CI
Give scientific reasons for the following:
Atomic radius goes on decreasing while going from left to right in a period.
Fill in the blanks.
Size of the atoms ______ from left to right across a period and ______ on descending in a group of normal elements.
Select the correct answer
Atomic radii of fluorine and neon in angstrom unit are
Nanometer unit is used to measure atomic radius.
Which of the following is the correct order of size?
Which of the following gives the correct increasing order of the atomic radii of O, F, and N?
Which of the following gives the correct increasing order of the atomic radii of O, F and N?
Which among the following elements has the largest atomic radii?
Which of the following are the characteristics of isotopes of an element?
- Isotopes of an element have same atomic masses
- Isotopes of an element have same atomic number
- Isotopes of an element show same physical properties
- Isotopes of an element show same chemical properties
Atomic number of a few elements are given below
10, 20, 7, 14
- Identify the elements
- Identify the Group number of these elements in the Periodic Table
- Identify the Periods of these elements in the Periodic Table
- What would be the electronic configuration for each of these elements?
- Determine the valency of these elements
- Electropositive nature of the element(s) increases down the group and decreases across the period
- Electronegativity of the element decreases down the group and increases across the period
- Atomic size increases down the group and decreases across a period (left to right)
- Metallic character increases down the group and decreases across a period.
On the basis of the above trends of the Periodic Table, answer the following about the elements with atomic numbers 3 to 9.
- Name the most electropositive element among them
- Name the most electronegative element
- Name the element with smallest atomic size
- Name the element which is a metalloid
- Name the element which shows maximum valency.
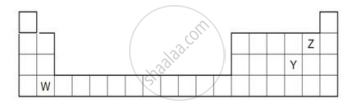
The diagram below shows part of the periodic table.
- Which elements would react together to form covalent compounds?
- Between the two elements W and Z, which will have a bigger atomic radius? Why?
This question refers to the elements of the Periodic Table with atomic numbers from 3 to 18. Some of the elements are shown by letters, but the letters are not the usual symbols of the elements.
| 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 |
| A | B | C | D | E | F | G | H |
| 11 | 12 | 13 | 14 | 15 | 16 | 17 | 18 |
| I | J | K | L | M | N | O | P |
Which of these have least atomic size in period 3?
