Advertisements
Advertisements
प्रश्न
Select the correct answer
Atomic radii of fluorine and neon in angstrom unit are
विकल्प
0.72, 13.6
1.60, 1.60
0.72, 0.72
none of these
Advertisements
उत्तर
0.72, 0.72
APPEARS IN
संबंधित प्रश्न
Which is greater in size an atom or an anion?
Which is greater in size Fe2+ or Fe3+?
Write the name and symbol of the element from the description.
The noble gas with the smallest atomic radius.
Among the elements of the second period, Li to Ne, pick out the element with the largest atomic size
Moving from left to right, the size of the atom decreases.
Which of the following is the correct order of atomic size?
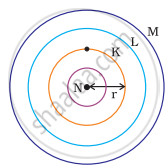
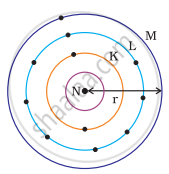
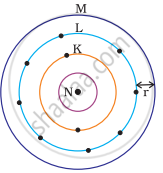
Which one of the following depict the correct representation of atomic radius(r) of an atom?
|
(i) |
(ii) |
|
(iii) |
(iv) |
Arrange the following as per the instruction given in the bracket:
Carbon, Fluorine, Beryllium (decreasing order of atomic size).
Arrange the following in order of increasing radii:
Mg2+, Mg, Mg+
Explain your choice.
Give reason for the following:
The size of a Cl− ion is greater than the size of a Cl atom.