Advertisements
Advertisements
प्रश्न
Write the name and symbol of the element from the description.
The noble gas with the smallest atomic radius.
Advertisements
उत्तर
Name: Helium
Symbol: He
संबंधित प्रश्न
Arrange the following as per the instruction given in the brackets:
Na, K, Li (Increasing atomic size)
Arrange the following as per the instruction given in the bracket:
Mg, Cl, Na, S, Si (decreasing order of atomic size).
What do you understand by atomic size? State its unit?
Give the trends in atomic size on moving down the group.
Why is the size of sodium is greater than magnesium?
Arrange the following in order of increasing radii:
CI- , CI
Write a scientific reason.
Atomic radius goes on increasing down a group.
Among the elements of the second period, Li to Ne, pick out the element with the largest atomic size
Fill in the blanks.
Size of the atoms ______ from left to right across a period and ______ on descending in a group of normal elements.
With reference to the variation of properties in the Periodic Table, which of the following is generally true?
Atomic size increases from left to right across a period.
Fill in the blank by selecting the correct word from the bracket.
If an element has seven electrons in its outermost shell then it is likely to have the _____ atomic size among all the elements in the same period.
The changes in the properties of elements on moving from left to right across a period of the Periodic Table. For the property, choose the correct answer.
The atomic size:
_______ is the distance between the nucleus of the atom and its outermost shell.
Write an Explanation.
Atomic radius
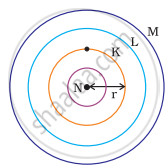
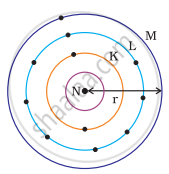
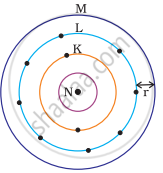
Which one of the following depict the correct representation of atomic radius(r) of an atom?
|
(i) |
(ii) |
|
(iii) |
(iv) |
An element X (atomic number 17) reacts with an element Y (atomic number 20) to form a divalent halide.
- Where in the periodic table are elements X and Y placed?
- Classify X and Y as metal (s), non-metal (s) or metalloid (s)
- What will be the nature of oxide of element Y? Identify the nature of bonding in the compound formed
- Draw the electron dot structure of the divalent halide
Arrange the following as per the instruction given in the bracket:
Carbon, Fluorine, Beryllium (decreasing order of atomic size).
In Period 3 of the Periodic Table, element B is placed to the left of element A. On the basis of this information, choose the correct word from the option to complete the following statement:
The element A would have ______ atomic size than B.
Give reason for the following:
The size of a Cl− ion is greater than the size of a Cl atom.