Advertisements
Advertisements
प्रश्न
Arrange the following in order of increasing radii:
Mg2+, Mg, Mg+
Explain your choice.
Advertisements
उत्तर
Mg2+< Mg+< Mg
Explanation:
Cation is smaller in size than its parent atom.
APPEARS IN
संबंधित प्रश्न
Arrange the following as per the instruction given in the bracket:
Mg, Cl, Na, S, Si (decreasing order of atomic size).
Arrange the elements of second and third periods in increasing order of their atomic size (excluding noble gases).
The size of an atom is indicated by its _______.
Nanometer unit is used to measure atomic radius.
Write information about the given atomic numbers in the table. 10, 20, 7.
| Atomic Number | Electronic configuration | Group | Period | Element |
| 10 | ||||
| 20 | ||||
| 7 |
Which among the following elements has the largest atomic radii?
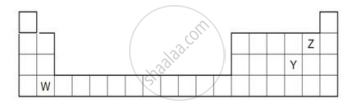
The diagram below shows part of the periodic table.
- Which elements would react together to form covalent compounds?
- Between the two elements W and Z, which will have a bigger atomic radius? Why?
Arrange the following in order of increasing radii:
N, O, P
Explain your choice.
This question refers to the elements of the Periodic Table with atomic numbers from 3 to 18. Some of the elements are shown by letters, but the letters are not the usual symbols of the elements.
| 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 |
| A | B | C | D | E | F | G | H |
| 11 | 12 | 13 | 14 | 15 | 16 | 17 | 18 |
| I | J | K | L | M | N | O | P |
Which of these have least atomic size in period 3?
