Advertisements
Advertisements
प्रश्न
Give reason for the following:
The size of a Cl− ion is greater than the size of a Cl atom.
Advertisements
उत्तर
An anion gets formed when one or more electrons are gained. Thus, there are more electrons than protons. The nucleus's effective positive charge is reduced, resulting in less inward pull. As a result, the size becomes larger.
\[\ce{\underset{(2, 8, 7)}{Cl}+e- ->\underset{(2, 8, 8)}{Cl-}}\]
APPEARS IN
संबंधित प्रश्न
Why is the size of neon greater than fluorine?
Study the radius of the element given below and answer the following questions.
| elements | K | Na | Rb | Cs | Li |
| Atomic radius (pm) | 231 | 186 | 244 | 262 | 151 |
a) Which of the above elements have the smallest atom?
b) In which group of the modern periodic table the above element are belongs?
c) What is the periodic trend observed in the variation of atomic radii down a group?
While going from top to bottom in a group the atomic radius _______.
Write information about the given atomic numbers in the table. 10, 20, 7.
| Atomic Number | Electronic configuration | Group | Period | Element |
| 10 | ||||
| 20 | ||||
| 7 |
Which of the following is the correct order of atomic size?
When an atom of iodine becomes an iodine ion (I–) the radius will ______
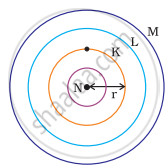
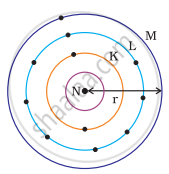
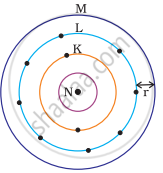
Which one of the following depict the correct representation of atomic radius(r) of an atom?
|
(i) |
(ii) |
|
(iii) |
(iv) |
Arrange the following as per instruction given in the bracket.
Mg, Cl, Na, S, Si (decreasing order of atomic size)
Arrange the following in order of increasing radii:
Cl−, Cl
Explain your choice.
Which one has the largest size?