Advertisements
Advertisements
प्रश्न
Arrange the following in increasing order of property indicated
I, I+, I- (atomic size)
Advertisements
उत्तर
I+ < I < I-
Size of a cation is always smaller than the corresponding atom due to decrease in number of electrons and increase in effective nuclear charge i.e., greater force of attraction by the nucleus on the electrons.
Size of an anion is always more than the corresponding atom due to decrease in effective nuclear charge i.e., lesser force of attraction by the nucleus o the electrons.
APPEARS IN
संबंधित प्रश्न
Give the trends in atomic size on moving across the period left to right.
Size of atom progressively becomes smaller when we move from sodium (Na) to chlorine (CI) in the third period of the periodic table?
Write a scientific reason.
Atomic radius goes on increasing down a group.
Arrange the following as per the instruction given in the bracket.
Mg, Cl, Na, S, Si (increasing order of atomic size)
Moving from left to right, the size of the atom decreases.
The electronic configuration of metal A is 2, 8, 18, 1.
The metal A when exposed to air and moisture forms B a green layered compound. A with con. H2 SO4 forms C and D along with water. D is a gaseous compound. Find A, B, C, and D.
Carbon belongs to the second period and Group 14. Silicon belongs to the third period and Group 14. If the atomic number of carbon is 6, the atomic number of silicon is ______
Which of the following gives the correct increasing order of the atomic radii of O, F and N?
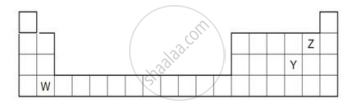
The diagram below shows part of the periodic table.
- Which elements would react together to form covalent compounds?
- Between the two elements W and Z, which will have a bigger atomic radius? Why?
In Period 3 of the Periodic Table, element B is placed to the left of element A. On the basis of this information, choose the correct word from the option to complete the following statement:
The element A would have ______ atomic size than B.
