Advertisements
Advertisements
प्रश्न
Write an Explanation.
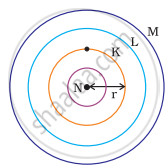
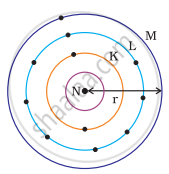
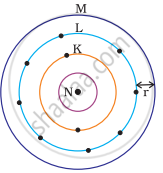
Atomic radius
Advertisements
उत्तर
Atomic radius is a measure of an atom’s size and changes predictably across periods and groups in the periodic table.
APPEARS IN
संबंधित प्रश्न
Arrange the following as per the instruction given in the brackets:
Na, K, Li (Increasing atomic size)
Answer the following in respect of element `31/15 P `
Give its formula with chlorine?
What do you understand by atomic size? State its unit?
Give the trends in atomic size on moving down the group.
Which is greater in size an atom or an anion?
Give reason for the following:
Argon atom is bigger than chlorine atom.
Write the name and symbol of the element from the description.
The noble gas with the smallest atomic radius.
The changes in the properties of elements on moving from left to right across a period of the Periodic Table. For the property, choose the correct answer.
The atomic size:
On moving from left to right in a periodic table, the size of the atom _______.
The size of an atom is indicated by its _______.
Atomic radius is expressed in the unit _______.
The size of an atom depends on the number of valence electrons.
Write scientific reason.
In same period, boron and oxygen elements have different atomic size.
Which of the following is the correct order of size?
Which one of the following depict the correct representation of atomic radius(r) of an atom?
|
(i) |
(ii) |
|
(iii) |
(iv) |
An element with the largest atomic radius among the following is ______.
Arrange the following as per instruction given in the bracket.
Mg, Cl, Na, S, Si (decreasing order of atomic size)
Arrange the following in order of increasing radii:
Mg2+, Mg, Mg+
Explain your choice.
Which one has the largest size?