Advertisements
Advertisements
प्रश्न
Write scientific reason.
In same period, boron and oxygen elements have different atomic size.
Advertisements
उत्तर
- Boron and oxygen are placed in the same period. However, their atomic numbers are different.
- Atomic number of boron is 5 and that of oxygen is 8.
- In a period, as the atomic number increases from boron to oxygen, the positive charge on the nucleus increases. At the same time, electrons get added in the same outermost shell. This increases effective nuclear charge.
- As a result, electrons are attracted closer to the nucleus decreasing the size of the oxygen atom.
Hence, in same period, boron and oxygen elements have different atomic size.
APPEARS IN
संबंधित प्रश्न
Give the trends in atomic size on moving down the group.
Which is greater in size an atom or a cation?
Which is greater in size Fe2+ or Fe3+?
Size of atom progressively becomes smaller when we move from sodium (Na) to chlorine (CI) in the third period of the periodic table?
Fill in the blank:
On moving across a period from right to left in the periodic table, the atomic size of the atom ___________.
Arrange the following in order of increasing radii:
CI- , CI
Write the name and symbol of the element from the description.
The noble gas with the smallest atomic radius.
Write a scientific reason.
Atomic radius goes on increasing down a group.
Study the radius of the element given below and answer the following questions.
| elements | K | Na | Rb | Cs | Li |
| Atomic radius (pm) | 231 | 186 | 244 | 262 | 151 |
a) Which of the above elements have the smallest atom?
b) In which group of the modern periodic table the above element are belongs?
c) What is the periodic trend observed in the variation of atomic radii down a group?
On moving from left to right in a periodic table, the size of the atom _______.
Atomic radius is expressed in the unit _______.
The electronic configuration of metal A is 2, 8, 18, 1.
The metal A when exposed to air and moisture forms B a green layered compound. A with con. H2 SO4 forms C and D along with water. D is a gaseous compound. Find A, B, C, and D.
Which of the following is the correct order of atomic size?
Which of the following is the correct order of atomic size?
Which among the following elements has the largest atomic radii?
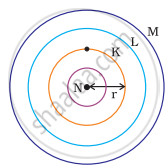
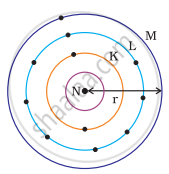
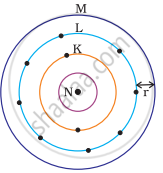
Which one of the following depict the correct representation of atomic radius(r) of an atom?
|
(i) |
(ii) |
|
(iii) |
(iv) |
Elements have been arranged in the following sequence on the basis of their increasing atomic masses.
| F, | Na, | Mg, | Al, | Si, | P, | S, | Cl, | Ar, | K |
- Pick two sets of elements which have similar properties.
- The given sequence represents which law of classification of elements?
Arrange the following in order of increasing radii:
N, O, P
Explain your choice.
Which one has the largest size?
Give reason for the following:
The size of a Cl− ion is greater than the size of a Cl atom.