Advertisements
Advertisements
प्रश्न
Write a scientific reason.
Atomic radius goes on increasing down a group.
Give scientific reasons:
Atomic radius goes on increasing down the ‘group’.
Advertisements
उत्तर
- Atomic radius increases as we move from top to bottom in a group because a new shell of electrons is added to the atoms at every step.
- The number of electron shells increases gradually, due to which the size of the atoms increases.
- As the size of the atoms increases, it leads to an increase in the atomic radius.
- The distance between the nucleus and the outermost electrons increases, so the attractive force of the nucleus decreases.
APPEARS IN
संबंधित प्रश्न
Arrange the following as per the instruction given in the brackets:
Na, K, Li (Increasing atomic size)
What do you understand by atomic size? State its unit?
Arrange the following in order of increasing radii:
CI- , CI
Study the radius of the element given below and answer the following questions.
| elements | K | Na | Rb | Cs | Li |
| Atomic radius (pm) | 231 | 186 | 244 | 262 | 151 |
a) Which of the above elements have the smallest atom?
b) In which group of the modern periodic table the above element are belongs?
c) What is the periodic trend observed in the variation of atomic radii down a group?
The following questions refer to the Periodic Table.
What happens to the atomic size of elements moving from top to bottom of a group?
Atomic radius is expressed in the unit _______.
While going from top to bottom in a group the atomic radius _______.
Nanometer unit is used to measure atomic radius.
Which of the following is the correct order of atomic size?
Which of the following is the correct order of size?
Which of the following gives the correct increasing order of the atomic radii of O, F, and N?
Which among the following elements has the largest atomic radii?
Which of the following are the characteristics of isotopes of an element?
- Isotopes of an element have same atomic masses
- Isotopes of an element have same atomic number
- Isotopes of an element show same physical properties
- Isotopes of an element show same chemical properties
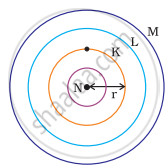
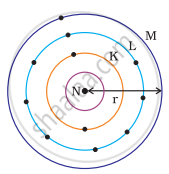
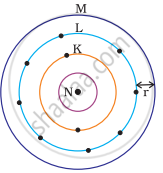
Which one of the following depict the correct representation of atomic radius(r) of an atom?
|
(i) |
(ii) |
|
(iii) |
(iv) |
An element X (atomic number 17) reacts with an element Y (atomic number 20) to form a divalent halide.
- Where in the periodic table are elements X and Y placed?
- Classify X and Y as metal (s), non-metal (s) or metalloid (s)
- What will be the nature of oxide of element Y? Identify the nature of bonding in the compound formed
- Draw the electron dot structure of the divalent halide
Arrange the following in order of increasing radii:
N, O, P
Explain your choice.
Which one of the following has the largest atomic radius?
Give reason for the following:
The size of a Cl− ion is greater than the size of a Cl atom.