Advertisements
Advertisements
प्रश्न
While going from top to bottom in a group the atomic radius _______.
पर्याय
increases
decreases
remains same
No change occurs
Advertisements
उत्तर
While going from top to bottom in a group the atomic radius increases.
APPEARS IN
संबंधित प्रश्न
Arrange the following as per the instruction given in the brackets:
Na, K, Li (Increasing atomic size)
Fill in the blank:
On moving across a period from right to left in the periodic table, the atomic size of the atom ___________.
Study the radius of the element given below and answer the following questions.
| elements | K | Na | Rb | Cs | Li |
| Atomic radius (pm) | 231 | 186 | 244 | 262 | 151 |
a) Which of the above elements have the smallest atom?
b) In which group of the modern periodic table the above element are belongs?
c) What is the periodic trend observed in the variation of atomic radii down a group?
What happens to the atomic size of elements on moving from left to right in a period?
The following questions refer to the Periodic Table.
What happens to the atomic size of elements moving from top to bottom of a group?
Supply the missing word from those in the brackets:
If an element has seven electrons in its outermost shell then it is likely to have the ______ (largest/ smallest) atomic size among all the elements in the same period.
The metals of Group 2 from top to bottom are Be, Mg, Ca, Sr and Ba. Which one of these elements will form ions most readily and why?
Arrange the following as per the instruction given in the bracket
Na, K, Li (Increasing atomic size)
The size of an atom is indicated by its _______.
Nanometer unit is used to measure atomic radius.
The electronic configuration of metal A is 2, 8, 18, 1.
The metal A when exposed to air and moisture forms B a green layered compound. A with con. H2 SO4 forms C and D along with water. D is a gaseous compound. Find A, B, C, and D.
Which of the following is the correct order of atomic size?
Which of the following gives the correct increasing order of the atomic radii of O, F, and N?
Which of the following are the characteristics of isotopes of an element?
- Isotopes of an element have same atomic masses
- Isotopes of an element have same atomic number
- Isotopes of an element show same physical properties
- Isotopes of an element show same chemical properties
Atomic number of a few elements are given below
10, 20, 7, 14
- Identify the elements
- Identify the Group number of these elements in the Periodic Table
- Identify the Periods of these elements in the Periodic Table
- What would be the electronic configuration for each of these elements?
- Determine the valency of these elements
- In below ladder symbols of elements are jumbled up. Rearrange these symbols of elements in the increasing order of their atomic number in the Periodic Table.
- Arrange them in the order of their group also.
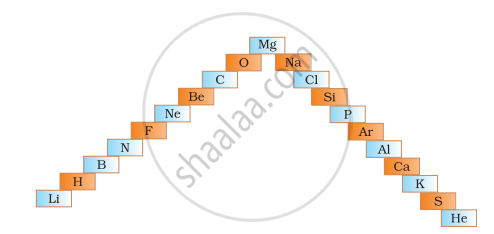
- Electropositive nature of the element(s) increases down the group and decreases across the period
- Electronegativity of the element decreases down the group and increases across the period
- Atomic size increases down the group and decreases across a period (left to right)
- Metallic character increases down the group and decreases across a period.
On the basis of the above trends of the Periodic Table, answer the following about the elements with atomic numbers 3 to 9.
- Name the most electropositive element among them
- Name the most electronegative element
- Name the element with smallest atomic size
- Name the element which is a metalloid
- Name the element which shows maximum valency.
Arrange the following as per the instruction given in the bracket:
Carbon, Fluorine, Beryllium (decreasing order of atomic size).
In Period 3 of the Periodic Table, element B is placed to the left of element A. On the basis of this information, choose the correct word from the option to complete the following statement:
The element A would have ______ atomic size than B.
