Advertisements
Advertisements
प्रश्न
Study the radius of the element given below and answer the following questions.
| elements | K | Na | Rb | Cs | Li |
| Atomic radius (pm) | 231 | 186 | 244 | 262 | 151 |
a) Which of the above elements have the smallest atom?
b) In which group of the modern periodic table the above element are belongs?
c) What is the periodic trend observed in the variation of atomic radii down a group?
Advertisements
उत्तर
a) Li
b) first group
c) while going down a group atomic radius goes on increasing. As a result, atomic size increases.
APPEARS IN
संबंधित प्रश्न
Answer the following in respect of element `31/15 P`
Give its electronic configuration
Answer the following in respect of element `31/15 P `
Is it a reducing agent or oxidizing agent?
Arrange the elements of second and third periods in increasing order of their atomic size (excluding noble gases).
Give reasons for the following:
The size of the anion is greater than the size of the parent atom.
Write a scientific reason.
Atomic radius goes on increasing down a group.
Supply the missing word from those in the brackets:
If an element has seven electrons in its outermost shell then it is likely to have the ______ (largest/ smallest) atomic size among all the elements in the same period.
Fill in the blank by selecting the correct word from the bracket.
If an element has seven electrons in its outermost shell then it is likely to have the _____ atomic size among all the elements in the same period.
Arrange the following as per the instruction given in the bracket
Na, K, Li (Increasing atomic size)
Some elements and their atomic radii are given here. Arrange them in decreasing order of their atomic radii. Identify which of the above elements is the biggest atom and which is smallest?
| Element | K | Na | Rb | Cs | Li |
| Atomic radius (pm) | 231 | 186 | 244 | 262 | 151 |
Write information about the given atomic numbers in the table. 10, 20, 7.
| Atomic Number | Electronic configuration | Group | Period | Element |
| 10 | ||||
| 20 | ||||
| 7 |
An element X has mass number 40 and contains 21 neutrons in its atom. To which group of the Periodic Table does it belong?
Which of the following is the correct order of size?
Which of the following is the correct order of atomic size?
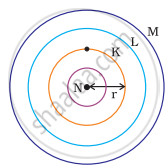
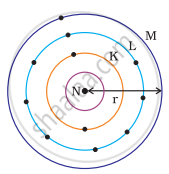
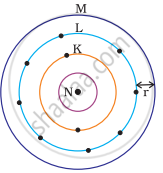
Which one of the following depict the correct representation of atomic radius(r) of an atom?
|
(i) |
(ii) |
|
(iii) |
(iv) |
Arrange the following elements in increasing order of their atomic radii.
- Li, Be, F, N
- Cl, At, Br I
Arrange the following as per the instruction given in the bracket:
Carbon, Fluorine, Beryllium (decreasing order of atomic size).
Arrange the following in order of increasing radii:
Cl−, Cl
Explain your choice.
Arrange the following in order of increasing radii:
Mg2+, Mg, Mg+
Explain your choice.
This question refers to the elements of the Periodic Table with atomic numbers from 3 to 18. Some of the elements are shown by letters, but the letters are not the usual symbols of the elements.
| 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 |
| A | B | C | D | E | F | G | H |
| 11 | 12 | 13 | 14 | 15 | 16 | 17 | 18 |
| I | J | K | L | M | N | O | P |
Which of these have least atomic size in period 3?