Advertisements
Advertisements
प्रश्न
Which is greater in size an atom or an anion?
Advertisements
उत्तर
An anion is greater than an atom since it is formed by the gain of electrons, and so the number of electrons is greater than the number of protons. The effective positive charge in the nucleus is less, so less inward pull is experienced. Hence, the size expands.
APPEARS IN
संबंधित प्रश्न
Answer the following in respect of element `31/15 P `
Give its formula with chlorine?
What do you understand by atomic size? State its unit?
Write a scientific reason.
Atomic radius goes on increasing down a group.
What happens to the atomic size of elements on moving from left to right in a period?
The changes in the properties of elements on moving from left to right across a period of the Periodic Table. For the property, choose the correct answer.
The atomic size:
The size of an atom is indicated by its _______.
Which of the following is the correct order of size?
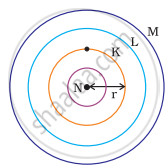
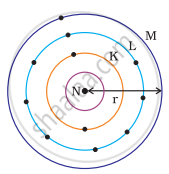
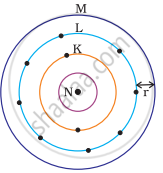
Which one of the following depict the correct representation of atomic radius(r) of an atom?
|
(i) |
(ii) |
|
(iii) |
(iv) |
Write the formula of the product formed when the element A (atomic number 19) combines with the element B (atomic number 17). Draw its electronic dot structure. What is the nature of the bond formed?
The diagram below shows part of the periodic table.
- Which elements would react together to form covalent compounds?
- Between the two elements W and Z, which will have a bigger atomic radius? Why?