Advertisements
Advertisements
Question
Which is greater in size an atom or an anion?
Advertisements
Solution
An anion is greater than an atom since it is formed by the gain of electrons, and so the number of electrons is greater than the number of protons. The effective positive charge in the nucleus is less, so less inward pull is experienced. Hence, the size expands.
APPEARS IN
RELATED QUESTIONS
Give the trends in atomic size on moving down the group.
Why is the size of neon greater than fluorine?
Write the name and symbol of the element from the description.
The noble gas with the smallest atomic radius.
The size of an atom is indicated by its _______.
Carbon belongs to the second period and Group 14. Silicon belongs to the third period and Group 14. If the atomic number of carbon is 6, the atomic number of silicon is ______
An element X has mass number 40 and contains 21 neutrons in its atom. To which group of the Periodic Table does it belong?
Which of the following is the correct order of atomic size?
- In below ladder symbols of elements are jumbled up. Rearrange these symbols of elements in the increasing order of their atomic number in the Periodic Table.
- Arrange them in the order of their group also.
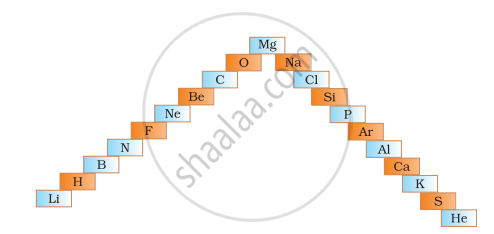
Arrange the following as per the instruction given in the bracket:
Carbon, Fluorine, Beryllium (decreasing order of atomic size).
In Period 3 of the Periodic Table, element B is placed to the left of element A. On the basis of this information, choose the correct word from the option to complete the following statement:
The element A would have ______ atomic size than B.
