Advertisements
Advertisements
Question
The size of an atom is indicated by its _______.
Options
atomic number
radius
number of shells
atomic mass
Advertisements
Solution
The size of an atom is indicated by its radius.
APPEARS IN
RELATED QUESTIONS
Answer the following in respect of element `31/15 P `
Is it a reducing agent or oxidizing agent?
Answer the following in respect of element `31/15 P `
Give its formula with chlorine?
Why is the size of neon greater than fluorine?
Which is greater in size an atom or a cation?
Which is greater in size Fe2+ or Fe3+?
Give scientific reasons for the following:
Atomic radius goes on decreasing while going from left to right in a period.
Select the correct answer
Atomic radii of fluorine and neon in angstrom unit are
The following questions refer to the Periodic Table.
What happens to the atomic size of elements moving from top to bottom of a group?
Supply the missing word from those in the brackets:
If an element has seven electrons in its outermost shell then it is likely to have the ______ (largest/ smallest) atomic size among all the elements in the same period.
The changes in the properties of elements on moving from left to right across a period of the Periodic Table. For the property, choose the correct answer.
The atomic size:
Atomic radius is expressed in the unit _______.
Nanometer unit is used to measure atomic radius.
Write information about the given atomic numbers in the table. 10, 20, 7.
| Atomic Number | Electronic configuration | Group | Period | Element |
| 10 | ||||
| 20 | ||||
| 7 |
Which of the following is the correct order of atomic size?
When an atom of iodine becomes an iodine ion (I–) the radius will ______
Which among the following elements has the largest atomic radii?
Elements have been arranged in the following sequence on the basis of their increasing atomic masses.
| F, | Na, | Mg, | Al, | Si, | P, | S, | Cl, | Ar, | K |
- Pick two sets of elements which have similar properties.
- The given sequence represents which law of classification of elements?
Arrange the following elements in increasing order of their atomic radii.
- Li, Be, F, N
- Cl, At, Br I
- In below ladder symbols of elements are jumbled up. Rearrange these symbols of elements in the increasing order of their atomic number in the Periodic Table.
- Arrange them in the order of their group also.
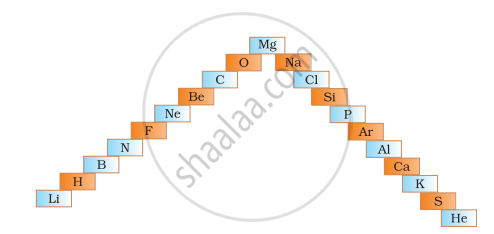
Arrange the following in order of increasing radii:
Cl−, Cl
Explain your choice.
